CHEM123L Study Guide - Final Guide: Ionic Strength, Intermolecular Force, Pipette
Document Summary
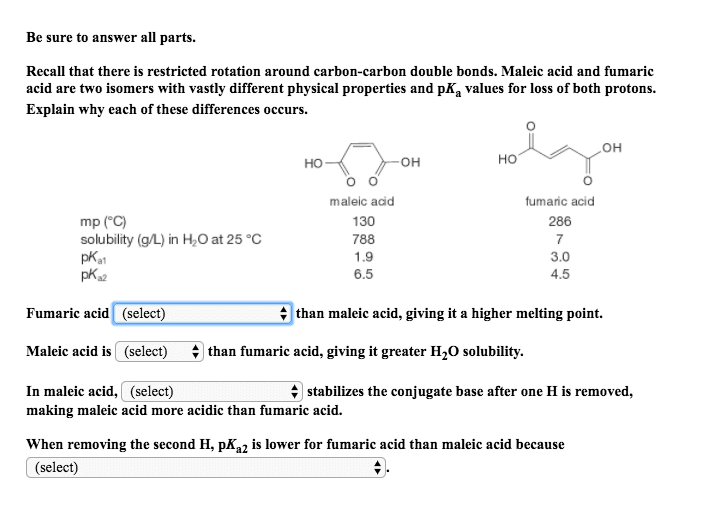
Sp3 (one 2s and three 2p), sp2 (one 2s and two 2p), sp (one 2s and one 2p) Experiment will produce and identify the cis and trans geometric isomers of 1,2- ethlenedicarboxylic acid; maleic acid (cis) and fumaric acid (trans) Intermolecular forces influenced melting point (fumaric higher than maleic) and solubility (fumaric insoluble, maleic soluble) Techniques used: quantitative transfer of samples and use of pipette. Know how to perform calculations from prelab (concentrations, ect) Know what happens to the rate constant when concentrations (no effect on doubling of product that does not have a keq; watch proportionalities), ionic strength (increases as strength decreases), and temperature is changed (increases rate as temperature increases) Know calculations for ionic strength (1/2 sum of (cizi)2), rate constant (rate = k [s2o8 and the rate (- s2o8. Experiment 3: weak acid-strong base and weak base-strong acid titrations. Techniques used: use of a burette in a titration and use of a volumetric pipette.