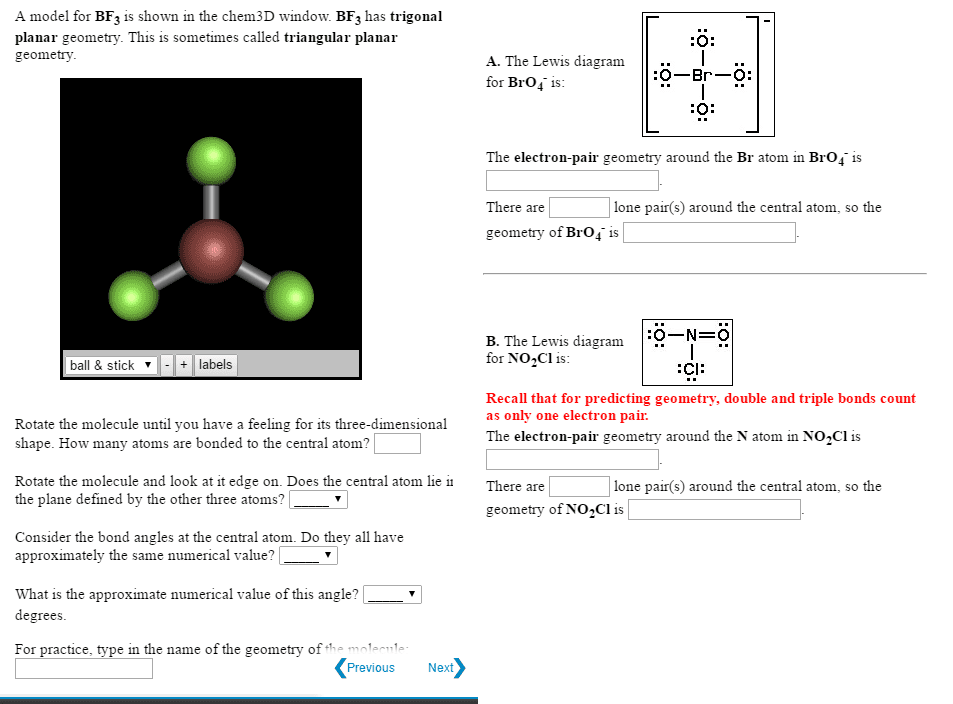
CHEM266 Study Guide - Midterm Guide: Lone Pair, Trigonal Planar Molecular Geometry, Molecular Orbital Theory
133 views5 pages
14 Dec 2016
School
Department
Course
Professor
Document Summary
Primary (1 ) is an sp3 c bonded to one other carbon. Secondary (2 ) is an sp3 c bonded to two other cs. Tertiary (3 ) is an sp3 c bonded to three other cs. Quaternary (4 ) is an sp3 c bonded to four other cs. Hydrogens take their classification from the c they are attached to. Double and triple bonds have higher priority than alkanes, halogens and nitro groups (but there are other higher priority groups, see below) Carbons involved in the double bond are trigonal planar and sp2 hybridized they have a bond angle of. ~120 , the double bond contains one bond and one bond (made up of two p orbitals) Carbons involved in the triple bond are linear and sp hybridized they have a bond angle of 180 , the triple bond contains one bond and two bonds.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232