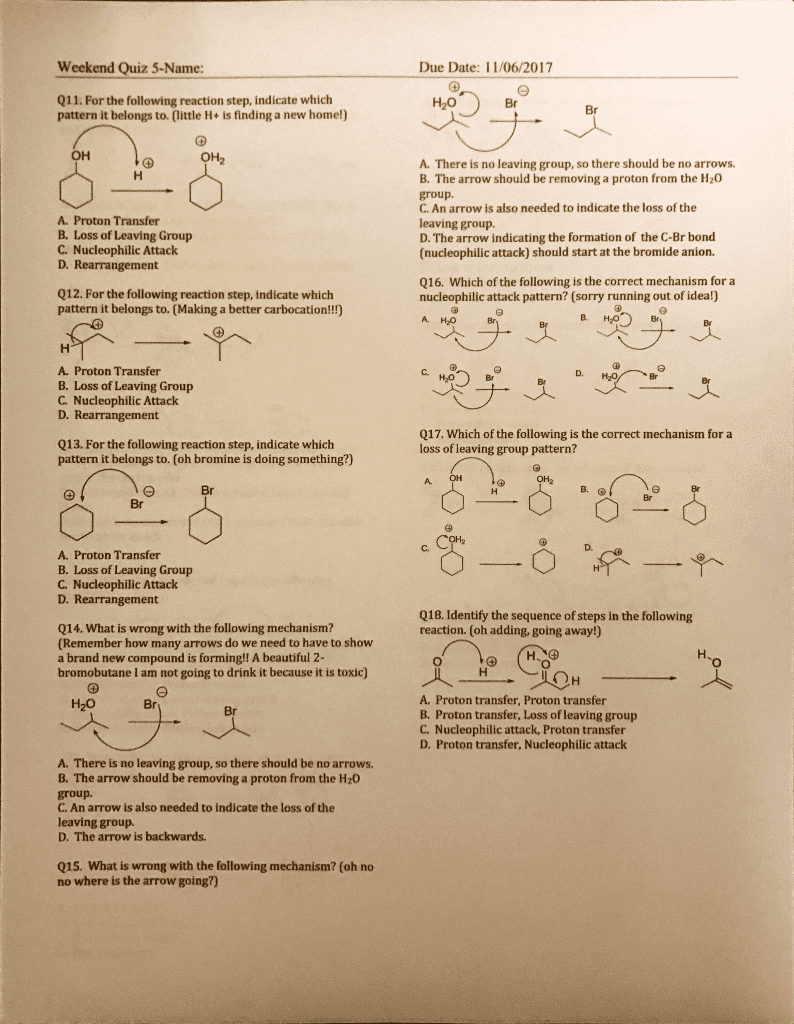
CHEM266 : Reaction Summary Table Including Mechanisms and Reactants, Products
Document Summary
Proton transfer to form most stable c+ (3 ) Carbocation, then water bonded to alkane: form carbocation from. Proton transfer to alkene: nucleophilic atk by water, proton transfer from water remove h from alkane. C, h bind to most subbed c: bond between b and bound h breaks, bh2 replace with oh. Hydration (addition favoured at lower temp but reagents present dominate temperature ) Rearrangement possible: formation of cyclo makes bigger delta positive charge on 3 c, anti addition, markovnikov selectivity, nu atk most subbed carbon, h added to least subbed c. Boron (big) bind to least subbed c (easier) Can form racemic pair enantiomers if chiral carbon form after. Syn addition can only add h and bh2 to top or bottom, limit # stereoisomers (must be cis) Can form meso compound: forms cyclo with br and 2 c, backside atk on more subbed c.