CHM 1311 Study Guide - Midterm Guide: Hypobromous Acid, Covalent Bond, Wave Function
Document Summary
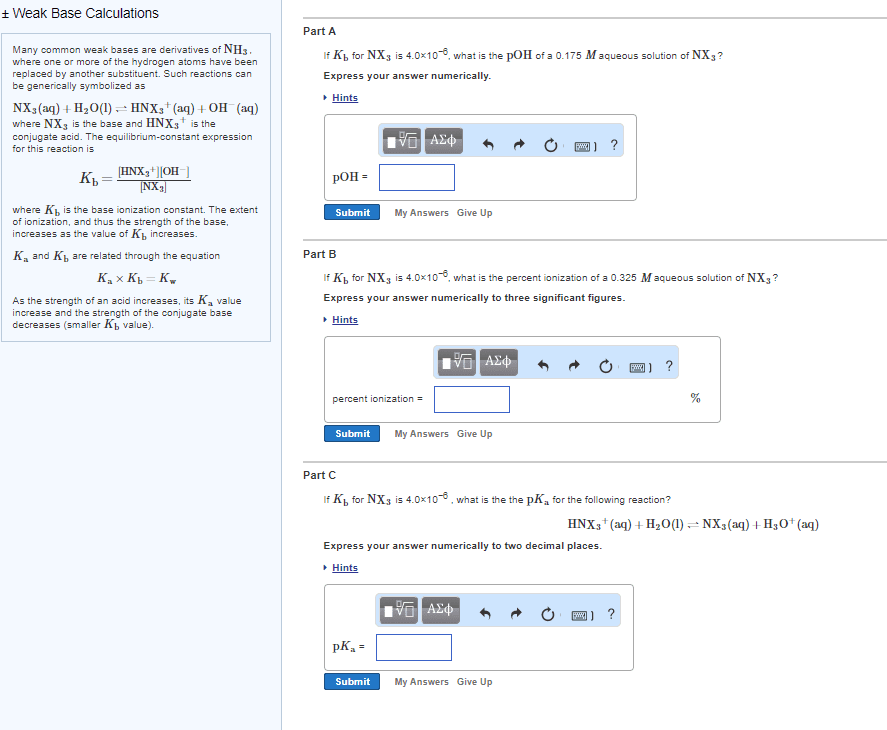
A 2. 85 g sample of a base symbolized by b, with a molar mass of 45. 09 g/mol, was dissolved in water to produce 100. 0 ml of solution with a ph = 11. 53. Write the equation for the reaction of b in water and calculate the ionization constant (kb) for this base. 2. 85g/45. 09g/mol = 6. 321 x 10-2; 0. 632m ph = 11. 53 poh = 14-11. 53 = -log [oh-] = 2. 47 [oh-] = 3. 388 x 10-3. E (3. 388 x 10-3)2/(0. 6286) = kb = 1. 826 x 10-5. The reaction corresponds to ka of the conjugate acid of base b. Calculated from the relationship kw = ka x kb for a conjugate pair. Thus ka = 10-14/1. 826 x 10-5 = 5. 47 x 10-10. Since 0. 66>>2x10-9 we will use the assumption and 0. 66-x = 0. 66 x2 = 2. 00x10-9 x 0. 660 = 1. 32x10-9 x = 3. 63 x 10-5. This is the [h+] so ph = 4. 44 (b) you now add 18. 51g of koh to this solution.