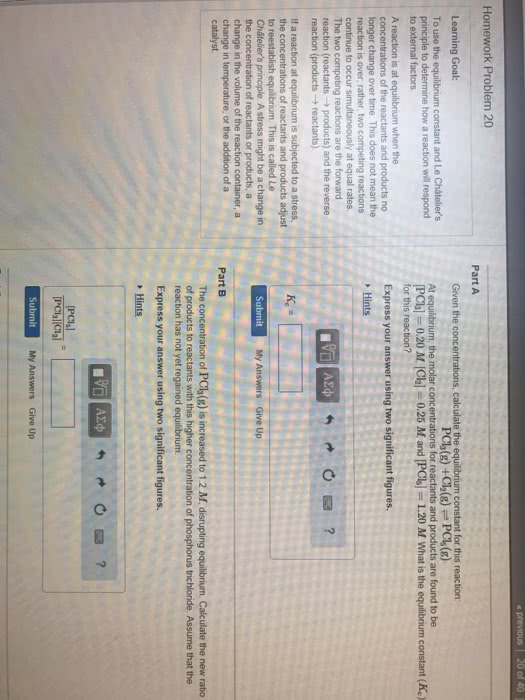
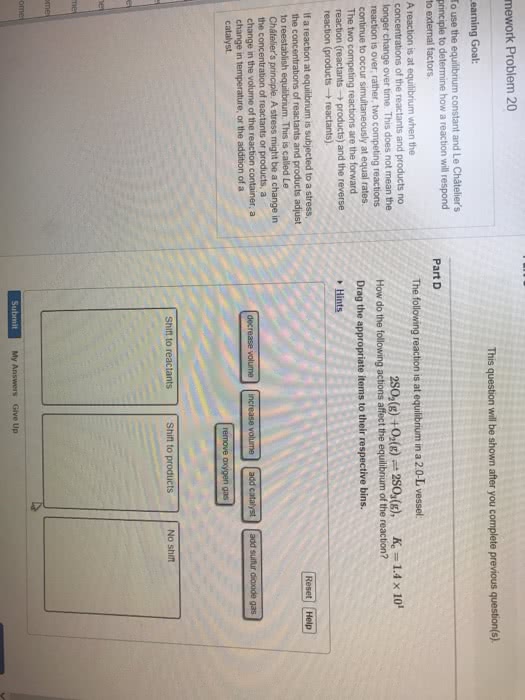
CHM 1311 : chem report 4.docx
Document Summary
Chemical kinetics studies how fast reactants change into products in a chemical reaction. It focuses on the reaction rate of an equation, the change in the amount of reactants or products over time. The rate varies between reactions and is determined from the nature of the reactants. Reactions such as explosions and the ripening of fruit occur over a different range of time. Since the reaction rate is given by the change in amount of reactants or products over time, we can derive the equation. Rate = change in concentration / change in time. If the concentration of the reactant decreases as the reaction occurs, then we put a negative sign around the rate equation to ensure it is positive. For the rate of an entire reaction with multiple reactants, the rate can be expressed as. This is called the rate law, and it expresses the rate as a function of the reactant (or product) concentrations.