

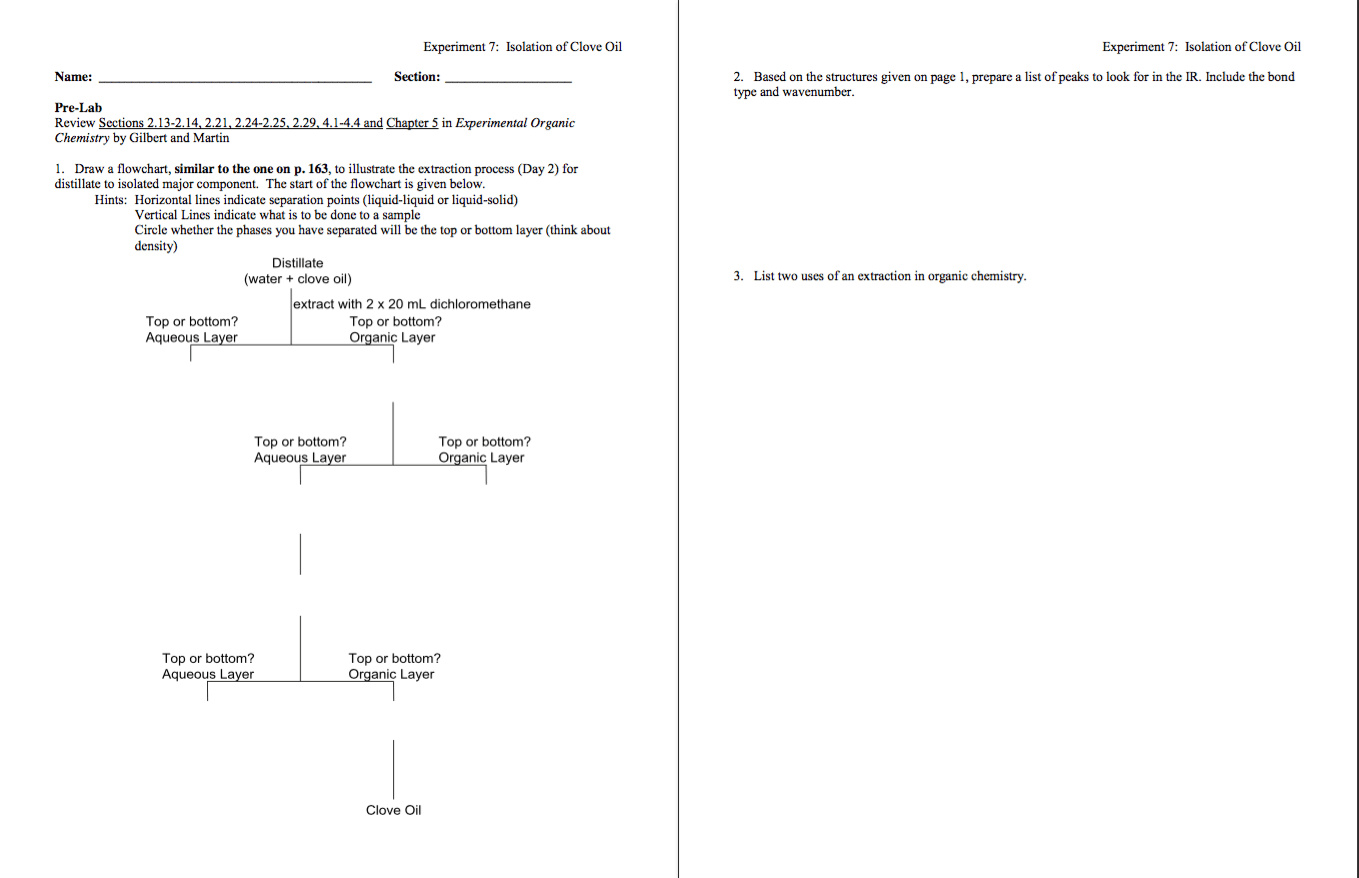
Wear safety glasses or goggles and suitable protective gloves while performing the experiments. Do not allow any of the chemicals used in this experiment t income in contact with your skin. If they do, immediately wash the affected areas with soap and water. Diethyl either is extremely volatile and flammable. Be certain there are no lames in your vicinity when using it for extraction and when removing it from the "Neutral Compound." Use caution in handingling wet glassware, because it can easily slip out of your grasp. When extracting a solution, vent the separatory funnel or vial often to avoid a buildup of pressure. Extract the clove oil from the distillate (saved on day 1) with two 20-mL portions of dichloromethane and combine the extracts. To do this...place the contents of the graduated cylinder and 20mL of dichloromethane in a closed separatory funnel. Cap the top with a glass stopper. Shake the contents to mix the layers then vent. Repeat the shaking/venting process 4-5 times. Allow the layers to separate. Drain the lower organic layer into a beaker. Add 20 mL of fresh dichloromethane to the separatory funnel then repeat the process. The two lower layers can be combined. In this extraction, the lower layer is the organic layer. The position of the layers depends on the densities of the two liquids involved. Because dichloromethane is halogenated, it has a density greater than 1, which makes it sink when combined with an aqueous solution having a density of about 1. You will need to be aware of the densities of liquids involved in later extractions to be able to track the layers. Extract the major component of clove oil from the dichloromethane solution obtained from the above extraction with two 15-mL portions of 1M sodium hydroxide (watch layers and think about what needs to be combined to get two layers in the separatory funnel). While monitoring with blue litmus paper, acidify the aqueous solution with 10 mL or more of 3M hydrochloric acid. Extract this aqueous solution with two 15-mL portions of dichloromethane and combine the organic extracts. Dry the dichloromethane solution from the previous paragraph with magnesium sulfate. Add small portions of magnesium sulfate to the dichloromethane solution. Magnesium sulfate clumps together as it picks up water from the solution. You have added enough when some of the magnesium sulfate is still freely moving in the dichloromethane solution. Adding too much will trap your product in the magnesium sulfate. Adding too little will leave large water impurities which we do not want for the IR at the end of lab. Carefully evaporate the dichloromethane using a pre-weighed flask on the rotavap (or by using a vacuum apparatus consisting of a pre-weighed, stoppered 50-mL filter flask heated on the lowest setting and a water trap cooled in an ice bath). When the boiling stops and the volume remains constant, weigh the product. Do not evaporate too much. Remember the product is volatile! Take your product and a clean and dry pipet to 346 to carry out the IR analysis, you may want to bring your graded pre-lab as well. You will be assisted in taking your IR, however, you will be expected to have a working knowledge next time. Pay attention! Mark the peaks that are given in your pre-lab list. Interpret your IR spectrum to prove the identity of the major component. You should be able to label enough peaks to prove beyond a shadow of a doubt what the main component is in your isolated clove oil. Draw a flowchart, similar lo the one on p. 163, to illustrate the extraction process (Day 2) for distillate to isolated major component. The start of the flowchart is given below. Based on the structures given on page 1, prepare a list of peaks to look for in the IR. Include the bond type and wavenumber. List two uses of an extraction in organic chemistry.







