CHM110H5 Study Guide - Final Guide: Stability Constants Of Complexes, Calibration Curve, Spectrophotometry
694 views10 pages
Document Summary
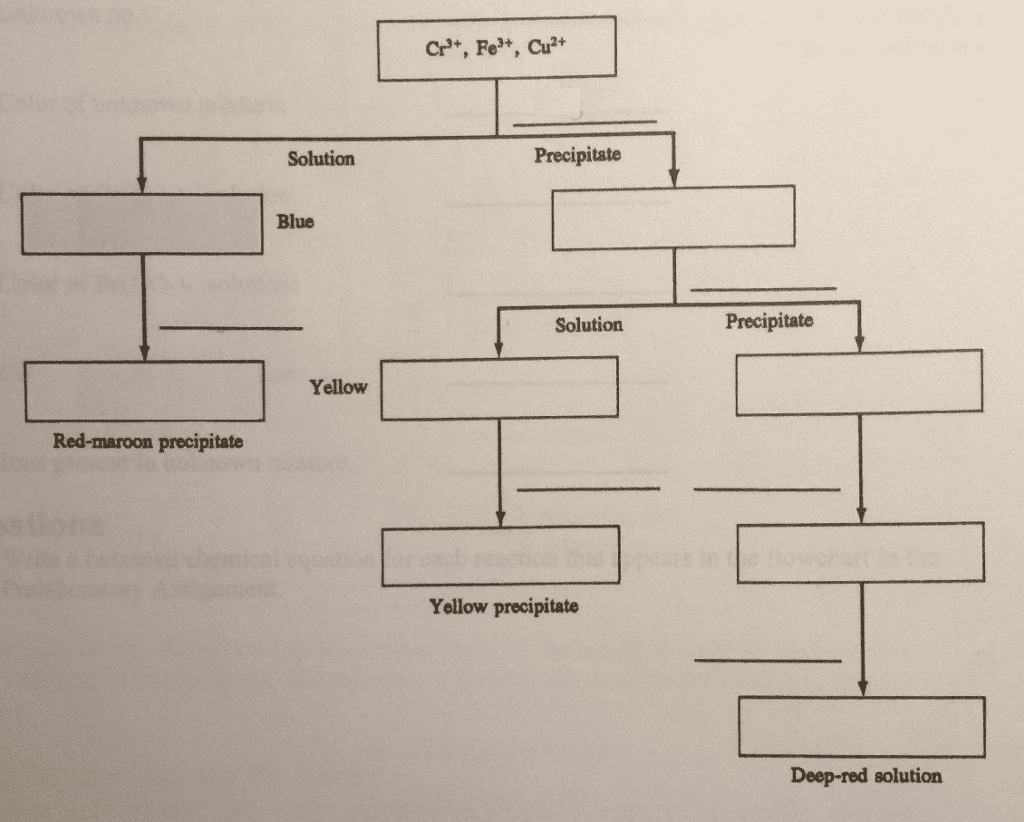
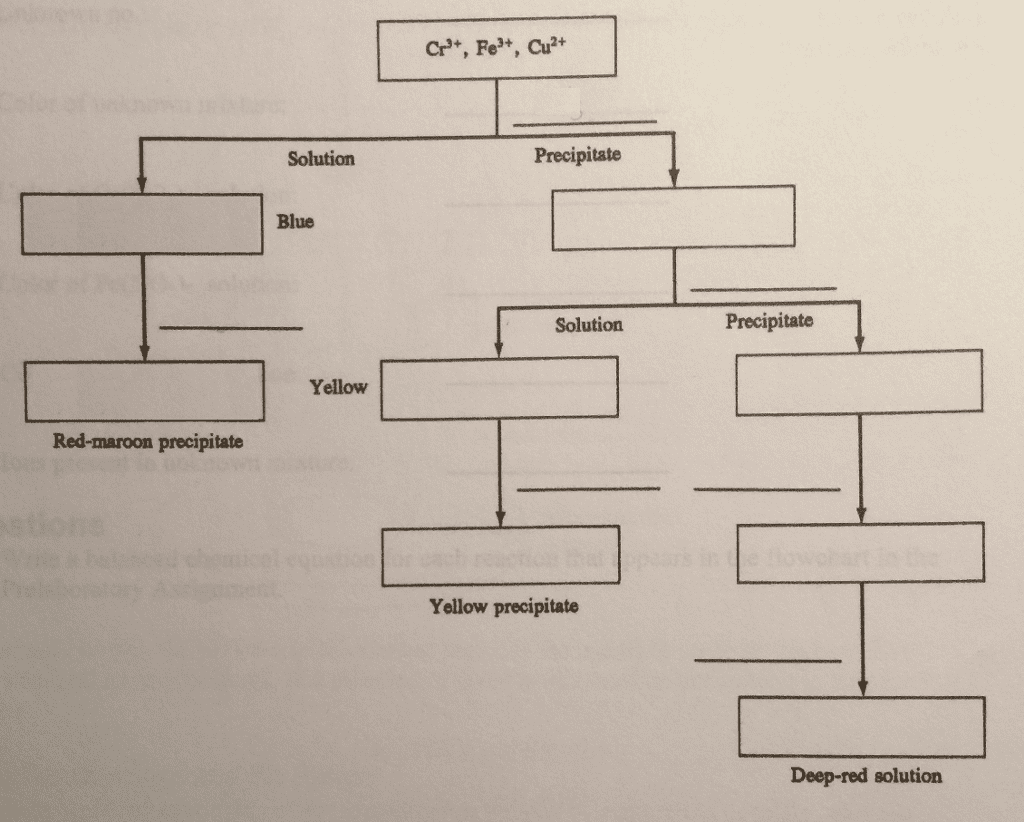
Iron ions can combine with ligands, molecules or ions with a lone pair of electrons, surrounding the metal ion to produce a very stable complex ion. All ligands have active lone pairs of electrons in their outermost energy level, which then form bonds with the central ion2. The stability of these complex ions is known as stability constants and can be written as the equilibrium constant for the formation of the complex ion. Some of the possible ligands ferric ions are able to react with are: h2o, nh3, cl-, co, cn= and scn-. In this work, the stability constant of the reaction between iron(iii) ions and thiocynate ions and its formation as a complex ion is determined with the use of several mixtures that have been mixed in different amounts. In this work, the solutions will be prepared by mixing known concentrations of hno3,
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers