CHMA10H3 Midterm: self test chp 1-6

36
CHMA10H3 Full Course Notes
Verified Note
36 documents
Document Summary
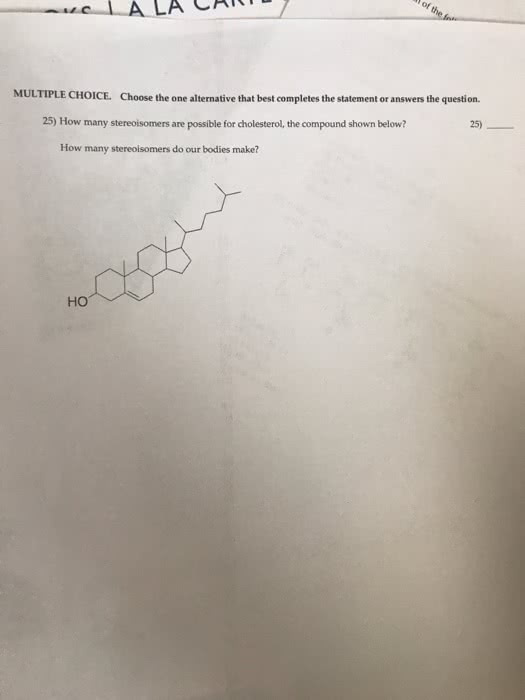
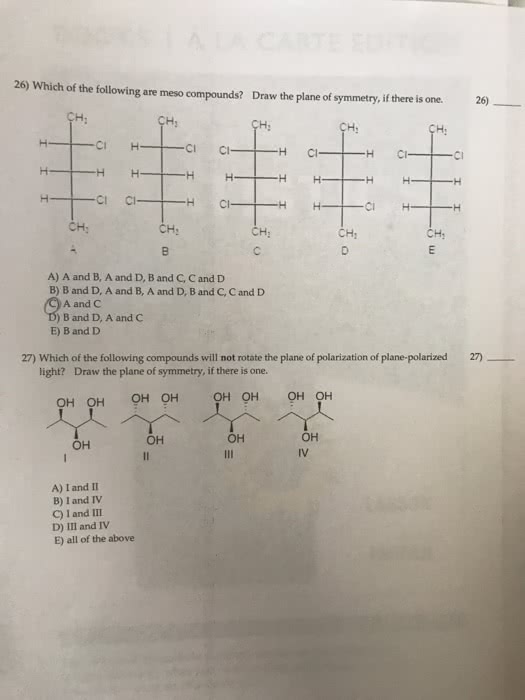
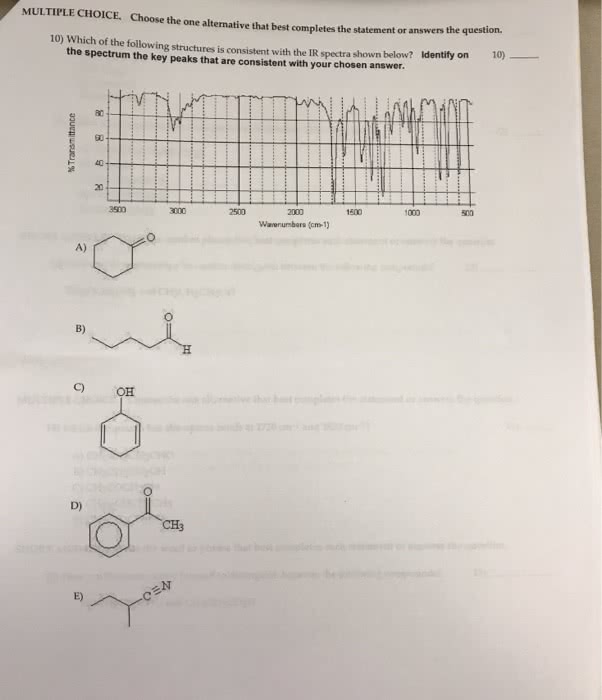
You should not take longer than 20 minutes to answer these 10 questions. Choose the one alternative that best completes the statement or answers the question: elements a and q form two compounds, aq and a2q. If it is a cation with a charge of +1, what is its correct symbol: 26. The only combustion products were 10. 942 g co2 and 4. 476 g h2o. Testname: chapter 13 review: 4, 6a, 1. 1, protons, neutrons, 2, 3, b, c, a, e, d, e www. notesolution. com. You should not take longer than30 minutes to answer these 10 questions. N2(g) + 3 h2(g) 2 nh3(g: hydrogen is the excess reactant. He took 4. 00 ml of the solution then diluted it with water to give 275 ml of a final solution. Testname: chapter 4 review: 13, limiting reactant, 0. 0684 m, d, d, c, c, c, b, d. Chma10h chapters 5 self assessment test 2008 www. notesolution. com.