CHM136H1 Study Guide - Final Guide: Dichlorocarbene, Lithium Aluminium Hydride, Dehydrohalogenation

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
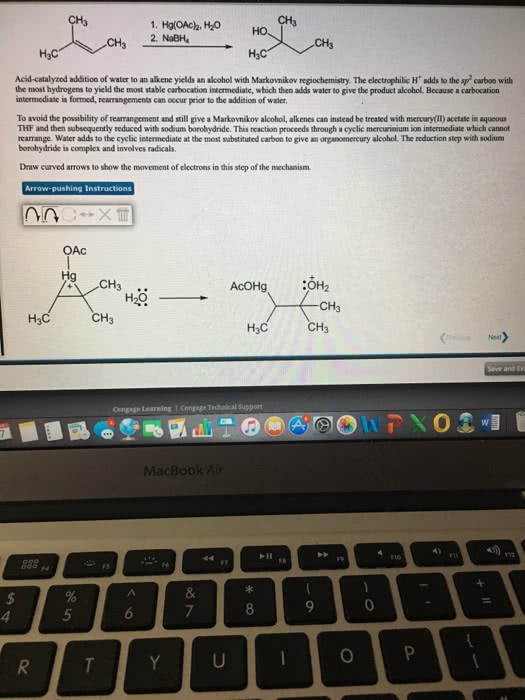
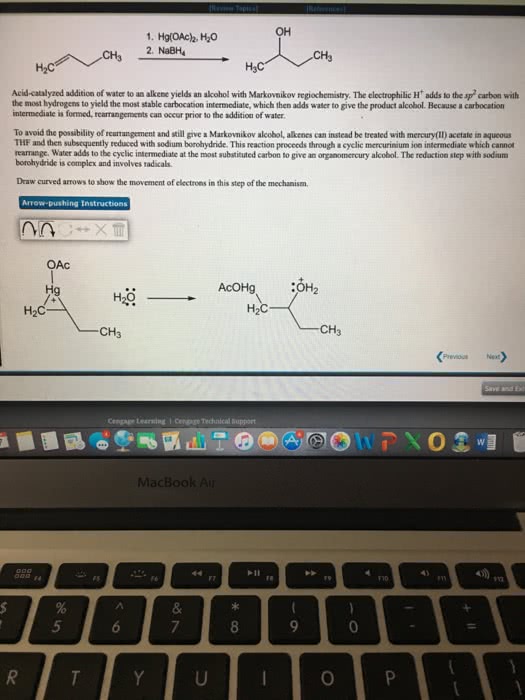
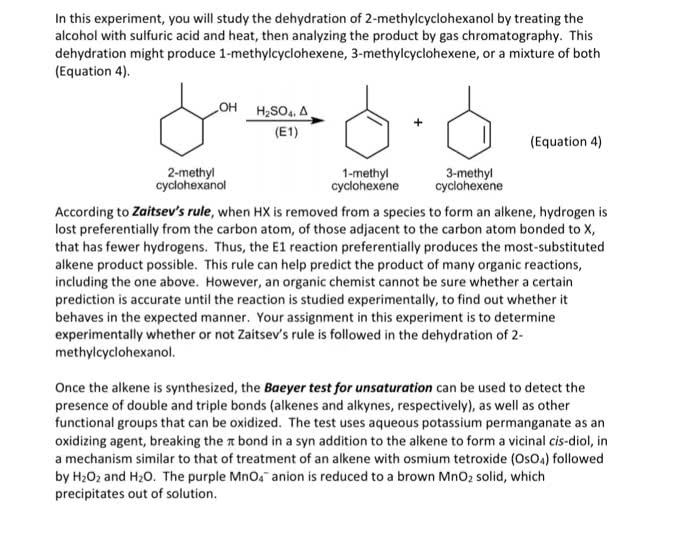
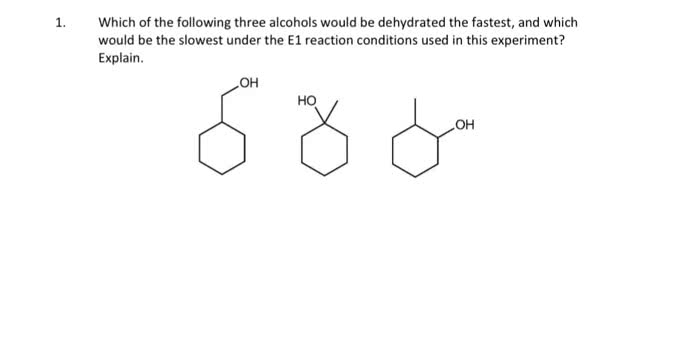
Electrophilic addition (to an alkene) hbr, hcl, and hi. Br2, cl2 in h2o aromatic rings do not react. Acid catalyzed hydration (only single double bonds reduced) 2 steps: double bond attacks h and electrons from hx bond go to x. Carbocation intermediate is formed (most stable/substituted: x anion attacks positive carbon follows markovnikov (halogen adds to more highly substituted carbon so it can have hydride and methyl shifts) 2 steps: double bond attacks one of the. Br and the 2 electrons from one of the br goes to the other. Bromonium cation intermediate formed: br attacks from other side. 2 steps: double bond attacks the br and the 2 electrons from one of the br goes to the other. Bromonium cation intermediate formed: h2o attacks from other side and makes positive o then water comes in and takes off the positive. 2 steps: double bond attacks the h3o+