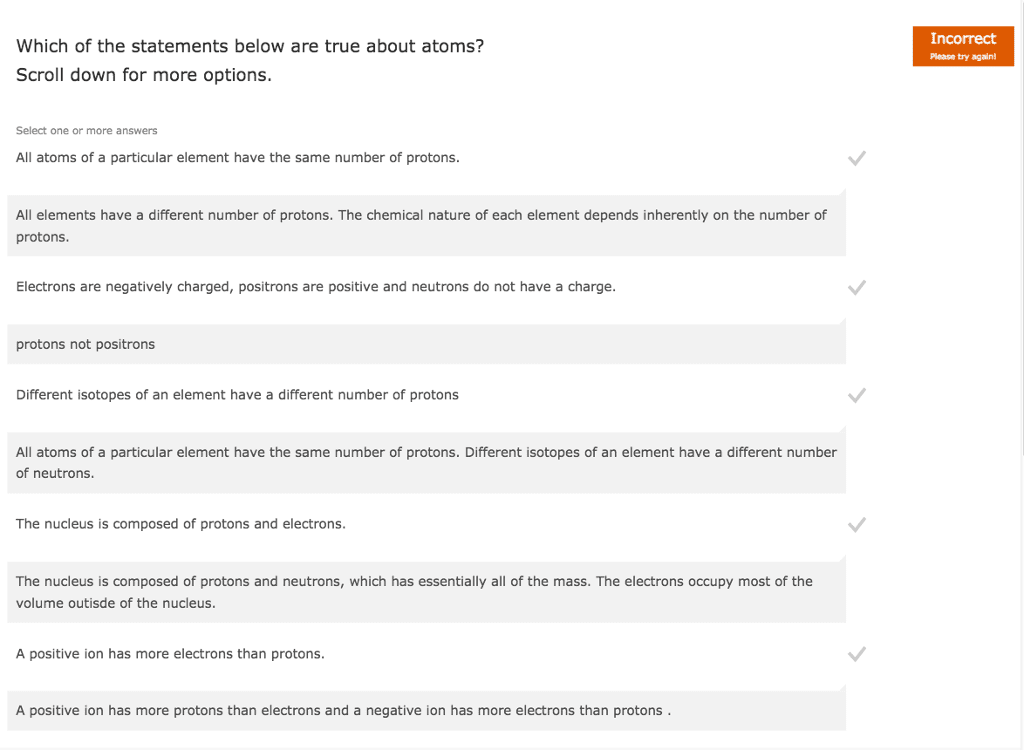
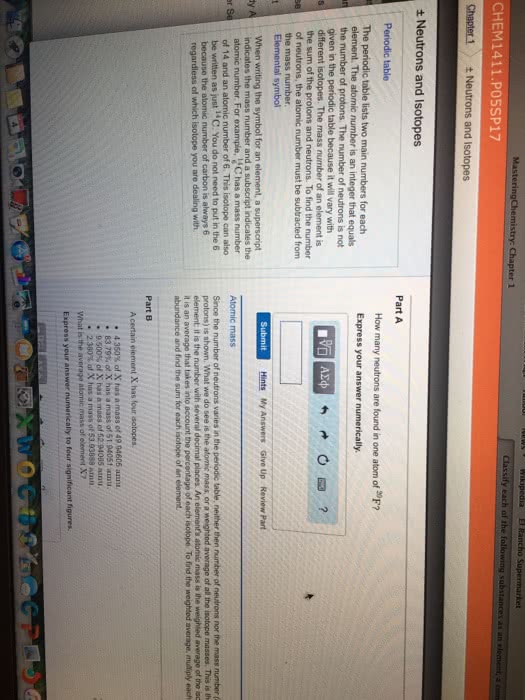
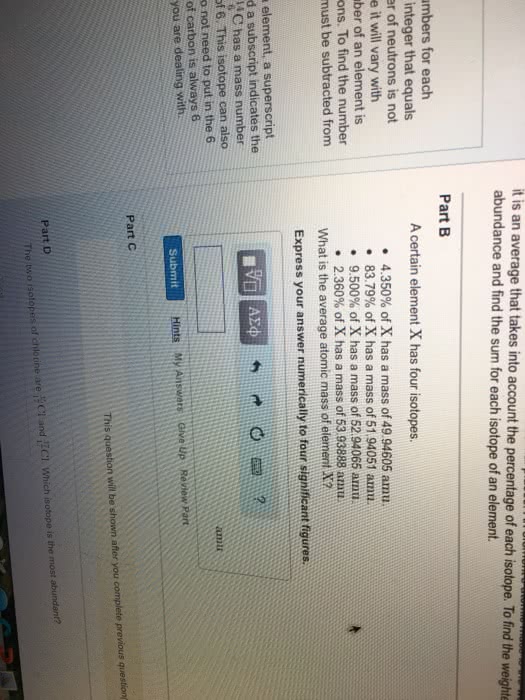
Wikipedia El Rancho Supermarket 1 Classify each of the following substances as an element, a cum CHEM 1411 P05SP17 Chapter 1 t Neutrons and Isotopes t Neutrons and Isotopes Part A Periodic table How many neutrons are foundin one atom of 20F? The periodic table lists two main numbers for each Express your answer numerically, element. The atomic number is an integer that equals n the number of protons. The number of neutrons is not given in the periodic table because it will vary with s different isotopes. The mass number of an element is the sum of the protons and neutrons. To find the number e of neutrons, the atomic number must be subtracted from the mass number. t Elemental symbol Submit Hints My Answers Give Up Review Part When writing the symbol for an element, a superscript indicates the mass number and a subscript indicates the num atomic number. For example, SC has a mass of 14 and an atomic number of 6. This isotope can also be written as just 14C. You do not need to put in the 6 Since the number of neutrons varies in the periodic table, neither then number of neutrons nor the mass number protons) is shown. What we do see is the atomic mass, or a weightod average of all the isotope masses. This is th because the atomic number of carbon is always 6 element it is the number with several decimal places. An element's adomie mass is the weighted average of the iso regardless of which isotope you are dealing with it is an average that takes into account the percentage of each isotope. To find tho weightod average. mutiply each abundance and find the sum for each isotope of an element. Part B A certain element X has four isotopes, 4350x of X has a mass of 49 94606 amu 83,79% of X has a mass of 51.94051 amu 9.500% of X has a mass of 52 94065 annu 2.360% of X has a mass of 53.93888 amta What is the average atomic mass of element X7 Express your answer numerically to four significant figures.