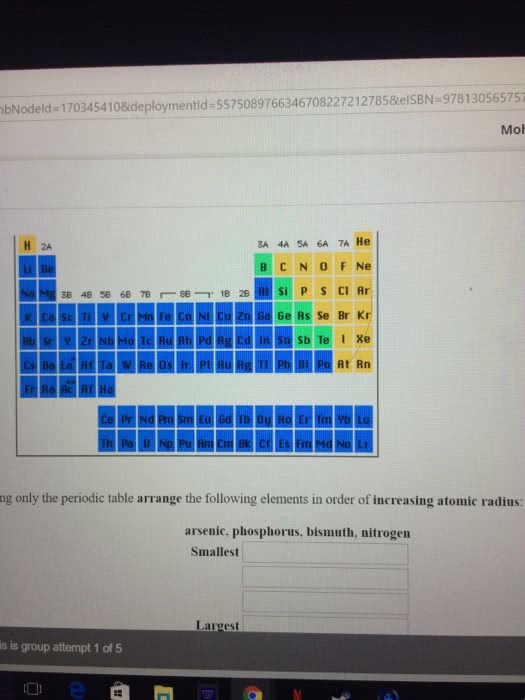
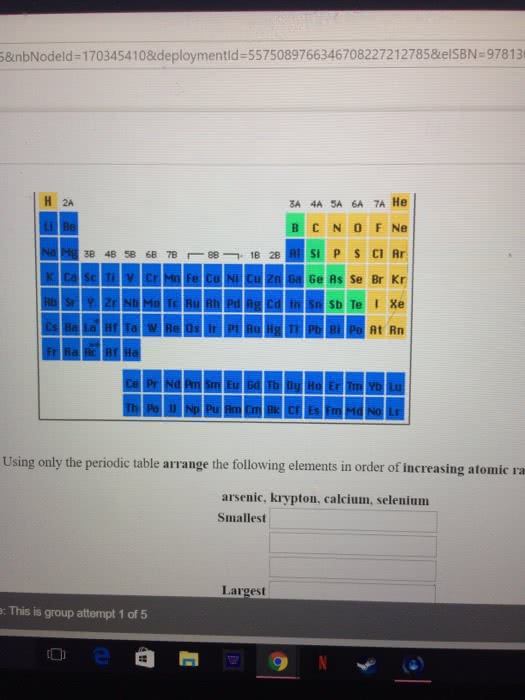
Are these correct? And how do you do #5 and #6?
Na M K Cal Sc Cr Mn Fe Co Ni Cu n Ga Ge As Se Br r cs | Ba | La | Hf | Ta w R@ Osl lr | Pt | Au | Hg ë Pb Ur Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu I. Which one of the following types of orbitals may be designated ywäth the quantum n -4,1-2,-+2? e) 4r ç±³2 depresentsthegrundstate of vanadium(atomp.323) pH's 'nnnnn.) 2. Which of the following 13 æ¯ 3. which is a valid spin-orbital diagram, for an atom in its ground state? A. B. âââ D.ââ ââ 2 E. 3 4) Which is an acceptable set of quantum numbers (n for a 3d electron? n ((A)),2,1+1/2 (B),í+1/2 (ST2 2 ã ¢-12 fD) 2 3 2-12 5) The number of unpaired electrons in the ground state of Mn i...(A)one (B) three (C) five (D) seven (D) seven Mn Is 6) The maximum number of electrons that can occupy an orbital labeled P, is... (A)I (B)2 (C)3 (D)4 7) Which of the C) 15 25 2p'2dD25'2p' E) None of the preceding choices 8) The configuration 4s2 3d o represents the two outer most (highest energy) sublevels in the ground state of an a o ng electron configurations represents the ground state of 0? A) Is2p' B) 122p'3 of (A) Mg (B) Ca (C) Fed) Zn (E) Cd 9 A Na lamp emits yellow light with a wavelength of 589 nm. What is the energy of a single photon in this light? 37 x 10 J. b. 1.12 x 10.c. 1.17 x 10J. d. 5.09 x 10'. e. 5.09 x 10'" .