CHEM 1001 Study Guide - Midterm Guide: Equivalence Point, Calcium Sulfite, Ph
Get access

Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
Calculate the pH of a 0.4310 M aqueous solution of sodium dihydrogen phosphate, NaH2PO4.
Use the Tables link on the toolbar for any equilibrium constants that are required
| Acid/Base Ionization Constants at 25 oC | ||||
|---|---|---|---|---|
| Acid | Formula | Ka1 | Ka2 | Ka3 |
| Acetic acid | CH3COOH | 1.8 Ã 10-5 | ||
| Acetylsalicylic acid (aspirin) | HC9H7O4 | 3.0 Ã 10-4 | ||
| Aluminum ion | Al(H2O)43+ | 1.2 Ã 10-5 | ||
| Arsenic acid | H3AsO4 | 2.5 Ã 10-4 | 5.6 Ã 10-8 | 3.0 Ã 10-13 |
| Ascorbic acid | H2C6H6O6 | 7.9 Ã 10-5 | 1.6 Ã 10-12 | |
| Benzoic acid | C6H5COOH | 6.3 Ã 10-5 | ||
| Carbonic acid | H2CO3 | 4.2 Ã 10-7 | 4.8 Ã 10-11 | |
| Ferric ion | Fe(H2O)63+ | 4.0 Ã 10-3 | ||
| Formic acid | HCOOH | 1.8 Ã 10-4 | ||
| Hydrocyanic acid | HCN | 4.0 Ã 10-10 | ||
| Hydrofluoric acid | HF | 7.2 Ã 10-4 | ||
| Hydrogen peroxide | H2O2 | 2.4 Ã 10-12 | ||
| Hydrosulfuric acid | H2S | 1.0 Ã 10-7 | 1.0 Ã 10-19 | |
| Hypochlorous acid | HClO | 3.5 Ã 10-8 | ||
| Nitrous acid | HNO2 | 4.5 Ã 10-4 | ||
| Oxalic acid | H2C2O4 | 5.9 Ã 10-2 | 6.4 Ã 10-5 | |
| Phenol | C6H5OH | 1.0 Ã 10-10 | ||
| Phosphoric acid | H3PO4 | 7.5 Ã 10-3 | 6.2 Ã 10-8 | 3.6 Ã 10-13 |
| Sulfuric acid | H2SO4 | very large | 1.2 Ã 10-2 | |
| Sulfurous acid | H2SO3 | 1.7 Ã 10-2 | 6.4 Ã 10-8 | |
| Zinc ion | Zn(H2O)42+ | 2.5 Ã 10-10 | ||
| Base | Formula | Kb | ||
|---|---|---|---|---|
| Ammonia | NH3 | 1.8 Ã 10-5 | ||
| Aniline | C6H5NH2 | 7.4 Ã 10-10 | ||
| Caffeine | C8H10N4O2 | 4.1 Ã 10-4 | ||
| Codeine | C18H21O3N | 8.9 Ã 10-7 | ||
| Diethylamine | (C2H5)2NH | 6.9 Ã 10-4 | ||
| Dimethylamine | (CH3)2NH | 5.9 Ã 10-4 | ||
| Ethylamine | C2H5NH2 | 4.3 Ã 10-4 | ||
| Hydroxylamine | NH2OH | 9.1 Ã 10-9 | ||
| Isoquinoline | C9H7N | 2.5 Ã 10-9 | ||
| Methylamine | CH3NH2 | 4.2 Ã 10-4 | ||
| Morphine | C17H19O3N | 7.4 Ã 10-7 | ||
| Piperidine | C5H11N | 1.3 Ã 10-3 | ||
| Pyridine | C5H5N | 1.5 Ã 10-9 | ||
| Quinoline | C9H7N | 6.3 Ã 10-10 | ||
| Triethanolamine | C6H15O3N | 5.8 Ã 10-7 | ||
| Triethylamine | (C2H5)3N | 5.2 Ã 10-4 | ||
| Trimethylamine | (CH3)3N | 6.3 Ã 10-5 | ||
| Urea | N2H4CO | 1.5 Ã 10-14 |
I've noticed that recently most of my chemistry questions that get answered have also been incorrect. I cant help but feel like this is a discrepncy between either the methods used, or the values of the refreances when choosing Ka,Ksp,Kb, costants of the nature. In order to avoid this I will post 2 pictures. The fisrt one will be the question I am asking, and the second one will be a worked question of similar nature that I missed. I will also post the refrences that I am using. Please use the numbers off those refernces or the answer will be wrong. Thanks
asking: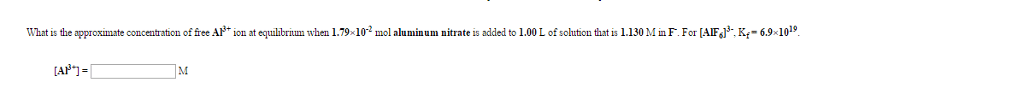
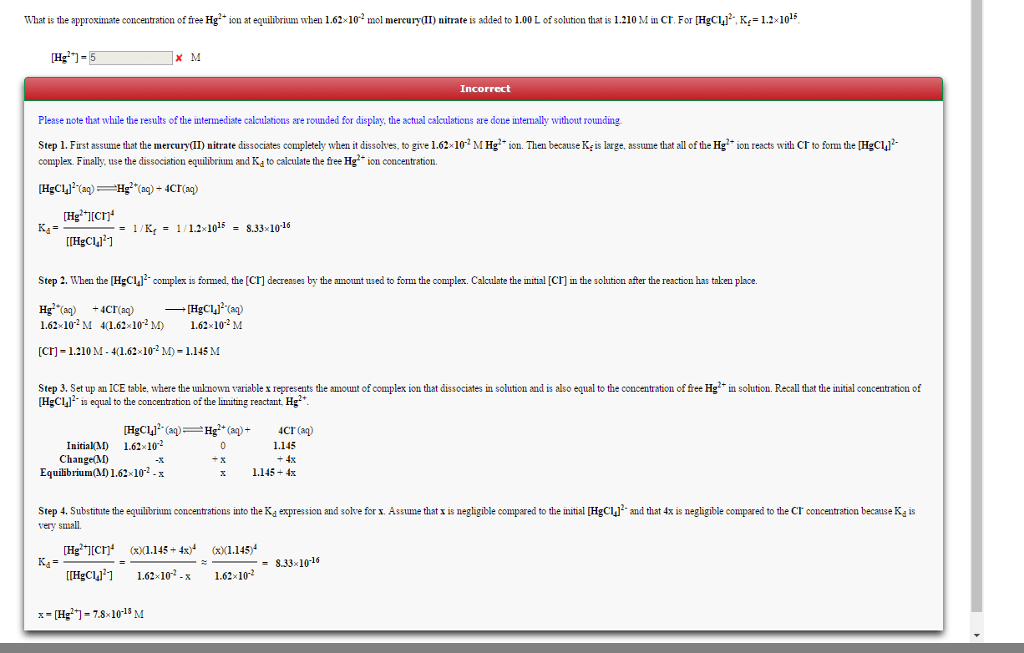
Charts:
Ksp-
BromidesPbBr26.3 Ã 10-6
AgBr3.3 Ã 10-13CarbonatesBaCO38.1 Ã 10-9
CaCO33.8 Ã 10-9
CoCO38.0 Ã 10-13
CuCO32.5 Ã 10-10
FeCO33.5 Ã 10-11
PbCO31.5 Ã 10-13
MgCO34.0 Ã 10-5
MnCO31.8 Ã 10-11
NiCO36.6 Ã 10-9
Ag2CO38.1 Ã 10-12
ZnCO31.5 Ã 10-11ChloridesPbCl21.7 Ã 10-5
AgCl1.8 Ã 10-10ChromatesBaCrO42.0 Ã 10-10
CaCrO47.1 Ã 10-4
PbCrO41.8 Ã 10-14
Ag2CrO49.0 Ã 10-12CyanidesNi(CN)23.0 Ã 10-23
AgCN1.2 Ã 10-16
Zn(CN)28.0 Ã 10-12FluoridesBaF21.7 Ã 10-6
CaF23.9 Ã 10-11
PbF23.7 Ã 10-8
MgF26.4 Ã 10-9HydroxidesAgOH2.0 Ã 10-8
Al(OH)31.9 Ã 10-33
Ca(OH)27.9 Ã 10-6
Cr(OH)36.7 Ã 10-31
Co(OH)22.5 Ã 10-16
Cu(OH)21.6 Ã 10-19
Fe(OH)27.9 Ã 10-15
Fe(OH)36.3 Ã 10-38
Pb(OH)22.8 Ã 10-16
Mg(OH)21.5 Ã 10-11
Mn(OH)24.6 Ã 10-14
Ni(OH)22.8 Ã 10-16
Zn(OH)24.5 Ã 10-17IodidesPbI28.7 Ã 10-9
AgI1.5 Ã 10-16OxalatesBaC2O41.1 Ã 10-7
CaC2O42.3 Ã 10-9
MgC2O48.6 Ã 10-5PhosphatesAlPO41.3 Ã 10-20
Ba3(PO4)21.3 Ã 10-29
Ca3(PO4)21.0 Ã 10-25
CrPO42.4 Ã 10-23
Pb3(PO4)23.0 Ã 10-44
Ag3PO41.3 Ã 10-20
Zn3(PO4)29.1 Ã 10-33SulfatesBaSO41.1 Ã 10-10
CaSO42.4 Ã 10-5
PbSO41.8 Ã 10-8
Ag2SO41.7 Ã 10-5SulfidesCaS8 Ã 10-6
CoS5.9 Ã 10-21
CuS7.9 Ã 10-37
FeS4.9 Ã 10-18
Fe2S31.4 Ã 10-88
PbS3.2 Ã 10-28
MnS5.1 Ã 10-15
NiS3.0 Ã 10-21
Ag2S1.0 Ã 10-49
ZnS2.0 Ã 10-25SulfitesBaSO38.0 Ã 10-7
CaSO31.3 Ã 10-8
Ag2SO31.5 Ã 10-14
Ka&Kb-
| Acetic acid | CH3COOH | 1.8 Ã 10-5 | ||
| Acetylsalicylic acid (aspirin) | HC9H7O4 | 3.0 Ã 10-4 | ||
| Aluminum ion | Al(H2O)43+ | 1.2 Ã 10-5 | ||
| Arsenic acid | H3AsO4 | 2.5 Ã 10-4 | 5.6 Ã 10-8 | 3.0 Ã 10-13 |
| Ascorbic acid | H2C6H6O6 | 7.9 Ã 10-5 | 1.6 Ã 10-12 | |
| Benzoic acid | C6H5COOH | 6.3 Ã 10-5 | ||
| Carbonic acid | H2CO3 | 4.2 Ã 10-7 | 4.8 Ã 10-11 | |
| Ferric ion | Fe(H2O)63+ | 4.0 Ã 10-3 | ||
| Formic acid | HCOOH | 1.8 Ã 10-4 | ||
| Hydrocyanic acid | HCN | 4.0 Ã 10-10 | ||
| Hydrofluoric acid | HF | 7.2 Ã 10-4 | ||
| Hydrogen peroxide | H2O2 | 2.4 Ã 10-12 | ||
| Hydrosulfuric acid | H2S | 1.0 Ã 10-7 | 1.0 Ã 10-19 | |
| Hypochlorous acid | HClO | 3.5 Ã 10-8 | ||
| Nitrous acid | HNO2 | 4.5 Ã 10-4 | ||
| Oxalic acid | H2C2O4 | 5.9 Ã 10-2 | 6.4 Ã 10-5 | |
| Phenol | C6H5OH | 1.0 Ã 10-10 | ||
| Phosphoric acid | H3PO4 | 7.5 Ã 10-3 | 6.2 Ã 10-8 | 3.6 Ã 10-13 |
| Sulfuric acid | H2SO4 | very large | 1.2 Ã 10-2 | |
| Sulfurous acid | H2SO3 | 1.7 Ã 10-2 | 6.4 Ã 10-8 | |
| Zinc ion | Zn(H2O)42+ | 2.5 Ã 10-10 |
| Base | Formula | Kb | ||
|---|---|---|---|---|
| Ammonia | NH3 | 1.8 Ã 10-5 | ||
| Aniline | C6H5NH2 | 7.4 Ã 10-10 | ||
| Caffeine | C8H10N4O2 | 4.1 Ã 10-4 | ||
| Codeine | C18H21O3N | 8.9 Ã 10-7 | ||
| Diethylamine | (C2H5)2NH | 6.9 Ã 10-4 | ||
| Dimethylamine | (CH3)2NH | 5.9 Ã 10-4 | ||
| Ethylamine | C2H5NH2 | 4.3 Ã 10-4 | ||
| Hydroxylamine | NH2OH | 9.1 Ã 10-9 | ||
| Isoquinoline | C9H7N | 2.5 Ã 10-9 | ||
| Methylamine | CH3NH2 | 4.2 Ã 10-4 | ||
| Morphine | C17H19O3N | 7.4 Ã 10-7 | ||
| Piperidine | C5H11N | 1.3 Ã 10-3 | ||
| Pyridine | C5H5N | 1.5 Ã 10-9 | ||
| Quinoline | C9H7N | 6.3 Ã 10-10 | ||
| Triethanolamine | C6H15O3N | 5.8 Ã 10-7 | ||
| Triethylamine | (C2H5)3N | 5.2 Ã 10-4 | ||
| Trimethylamine | (CH3)3N | 6.3 Ã 10-5 | ||
| Urea | N2H4CO | 1.5 Ã 10-14 |

