CHEM 1001 Study Guide - Midterm Guide: Reaction Quotient, Weak Base
Document Summary
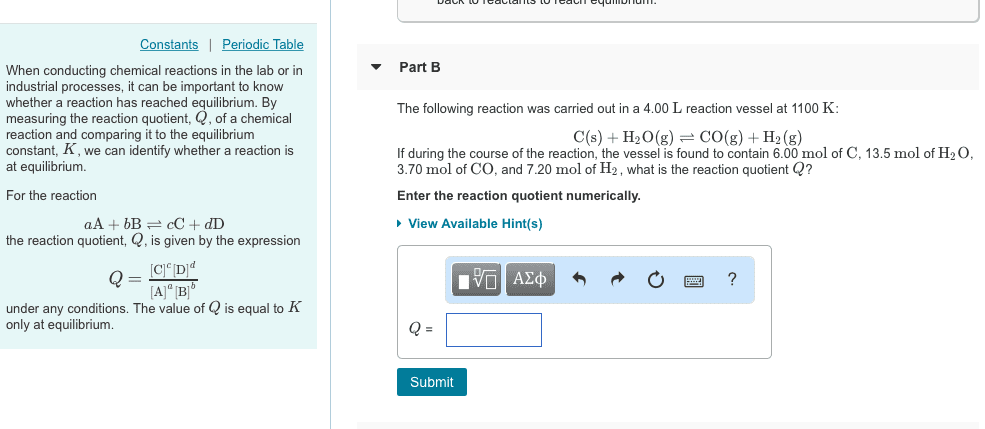
Quiz 2 section m 1 march 2012 (4) 1. A liquid reaction a + b c reached equilibrium in volume v1 (the equilibrium constant is known and equal to kc). This equilibrium mixture is diluted with the same solvent to a total volume of v2. A weak base b is dissolved in water at a known concentration cb. The ion product of water is known an equal to (kw= [h3o+][oh-]). Derive and expression for ph as a function of kw, cb and kb. Do not make any assumption about the degree of ionization. To simplify your solution, do not consider the contribution of water self-ionization: reaction a(g) + (b) c(g), h < 0. Will the equilibrium shift and which way will i shift if one of the following. Explain each of your answers using the le chatelier"s principle in no more than. Equimolar amounts of aqueous solutions of naoh and fh are mixed and allowed to react and reach equilibrium.