CHM 114 Study Guide - Midterm Guide: Reaction Rate, Reaction Rate Constant, Rate Equation
Document Summary
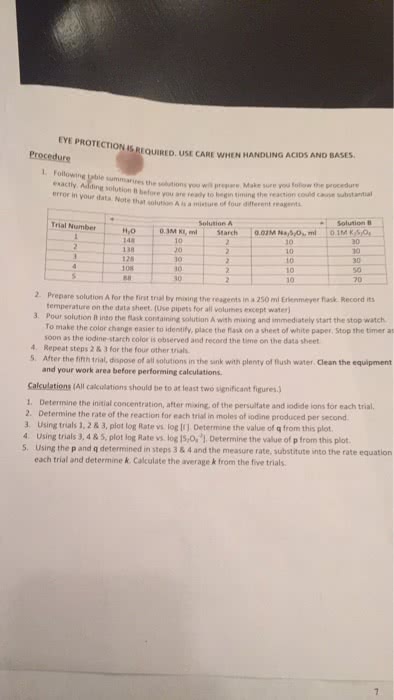
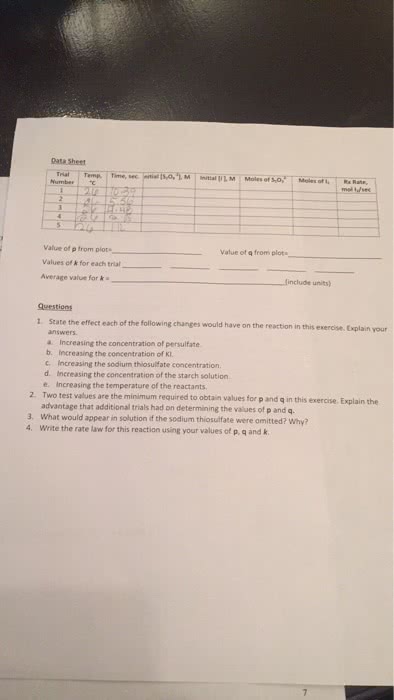
In contrast to the exams 1, 2 and 3, all having 25 questions each, this exam has 20 questions. Mark your answers and if relevant write your calculations on the questionnaire. Write and bubble your last and first name on scantron of corresponding color code. To understand what the reaction rate is (molarity change per time change; this is the rate of disappearance of a reactant and the rate of appearance of a product). To understand the effect of the concentration of reactants on the reaction rate. What is the reaction order in a certain reactant? (the power of magnitude at the concentration of the reactant). To remember that the reaction order can be fractional, it must be determined experimentally, although for simplicity we consider only 0, 1st and 2nd orders. To remember that unit of the rate constant depends on the reaction order (m/s for the zero, 1/s for the 1st and 1/(m s) for the 2nd order).