CHEM1161 : 4.6-end.docx
Document Summary
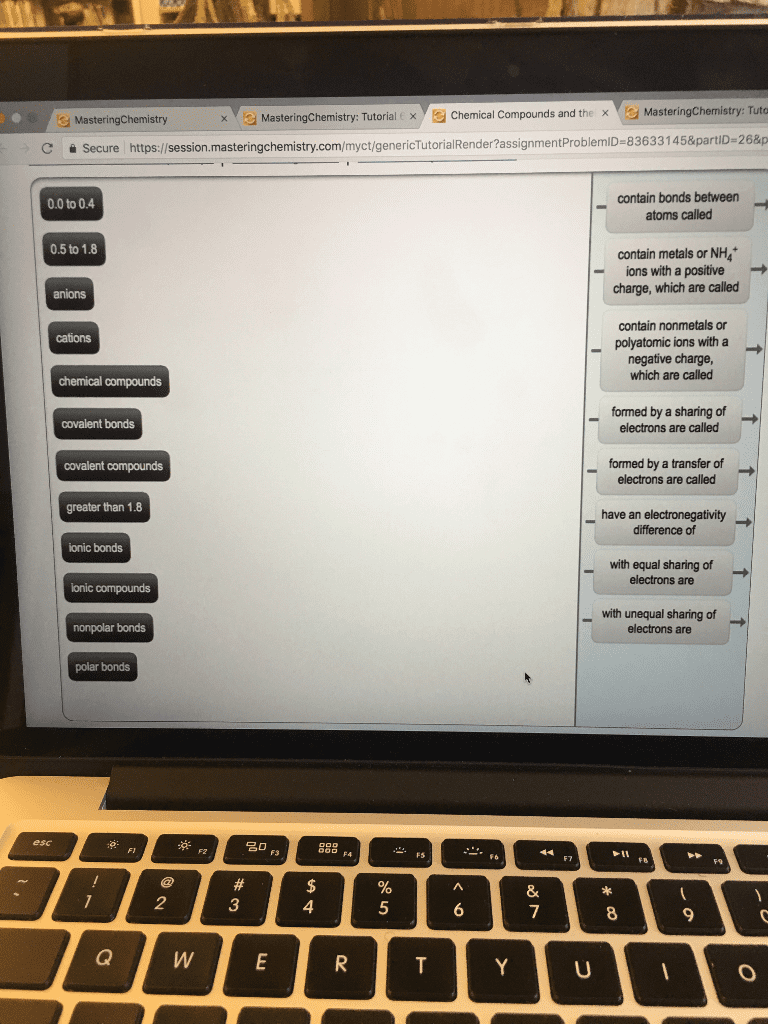
Of an element indicates the ability of an atom to attract the shared electrons in a. Increases from left to right going across a period. Is high for the nonmetals, with f as the highest ,cs has the lowest. A nonpolar covalent bond: occurs between nonmetals, is an equal or almost equal sharing of electrons, has a very small electronegativity difference. N-n 3. 0 3. 0 = 0. 0 nonpolar covalent. Cl-br 3. 0 2. 8 = 0. 2 nonpolar covalent. H-si 2. 1 1. 8 = 0. 3 nonpolar covalent. A polar covalent bond: occurs between nonmetal atoms, is an unequal sharing of electrons, has a moderate electronegativity difference. O-cl 3. 5 3. 0 = 0. 5 polar covalent. Cl-c 3. 0 2. 5 = 0. 5 polar covalent. O-s 3. 5 2. 5 = 1. 0 polar covalent. An ionic bond: occurs between metal and nonmetal ions, is a result of electron transfer, has a large electronegativity difference (1. 8 or more) Ex: pbr3 - p 5 e- shape: trigonal pyramidal.