CHEM 3311 Study Guide - Midterm Guide: Antibonding Molecular Orbital, Molecular Orbital Theory, Atomic Orbital
Document Summary
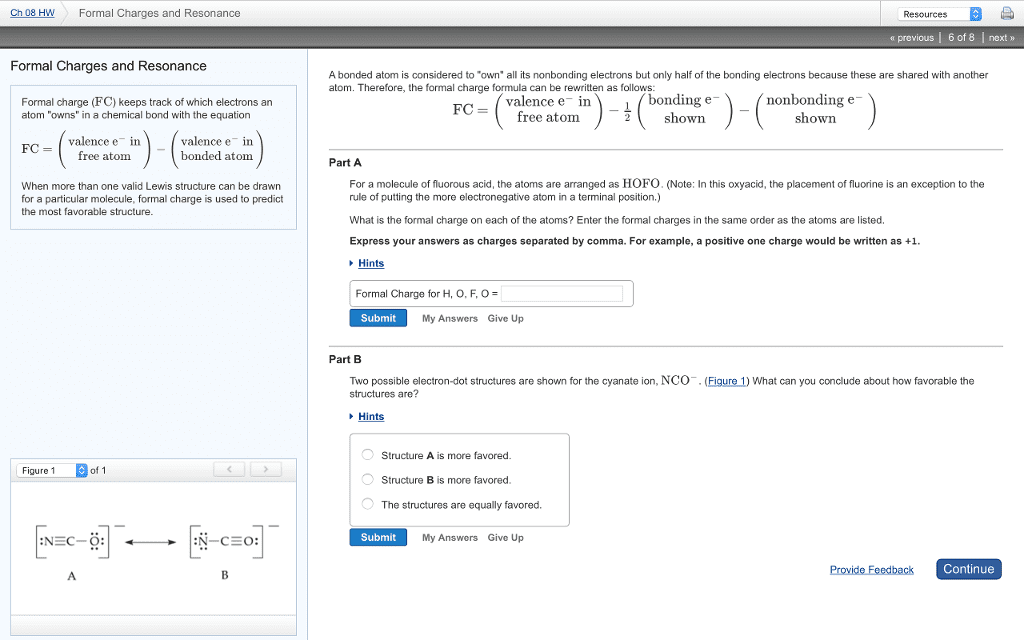
Set 1: lewis structures, resonance and functional groups. Formal charge: formal charge = valence e (nonbonding e + bonds: curved arrow notation: Shows the movement of electrons, either real (in a rxn) or imaginary (resonance) In the case of resonance, it is imaginary movement electrons are not moving, they are delocalizing/spreading out over a larger area: assessing contributors to the resonance hybrid, octet rule: most important! Functional groups: a functional group is a group of atoms (or a single atom) in an organic molecule that has a characteristic reactivity (way of reacting, hydrocarbons (contain c, h only): Organic chemistry midterm study guide: aliphatic hydrocarbons, alkanes: hydrocarbon it all single bonds, all carbons have sp3 hybridization, saturated hydrocarbon , classification: 1 (primary) carbon attached to 1 c. 2 (secondary) carbon attached to 2 c. 3 (tertiary) carbon attached to 3 c.