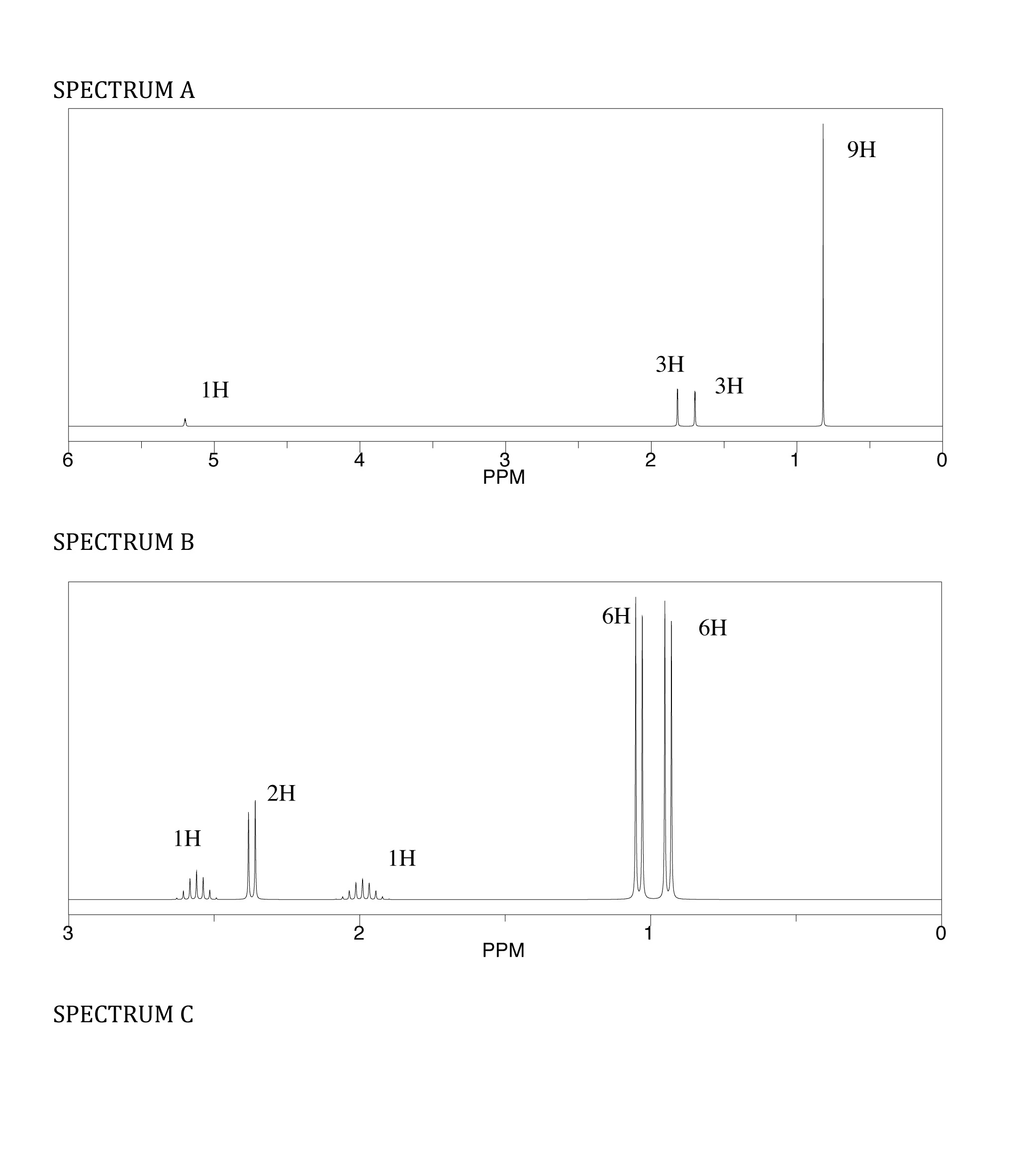
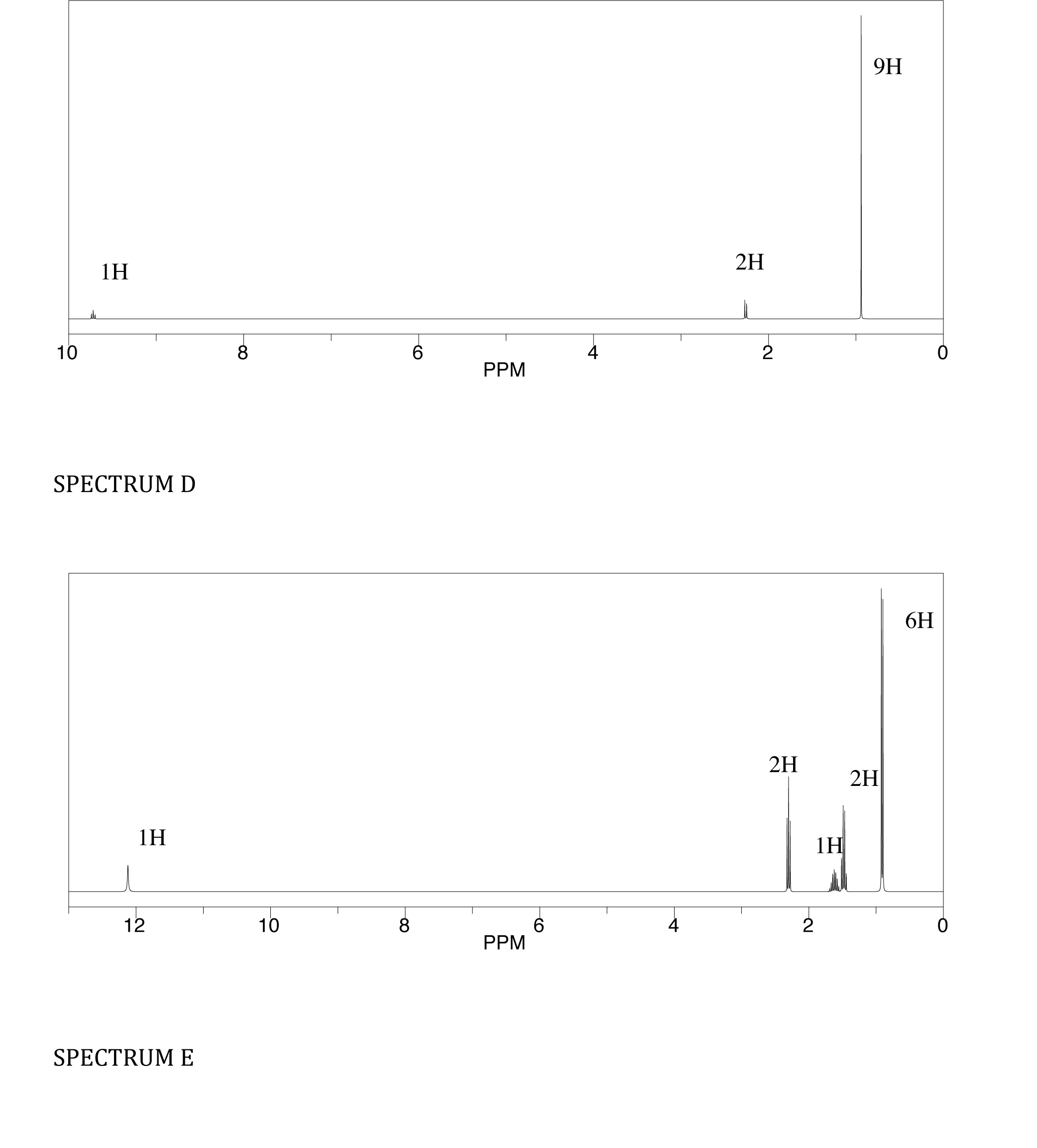
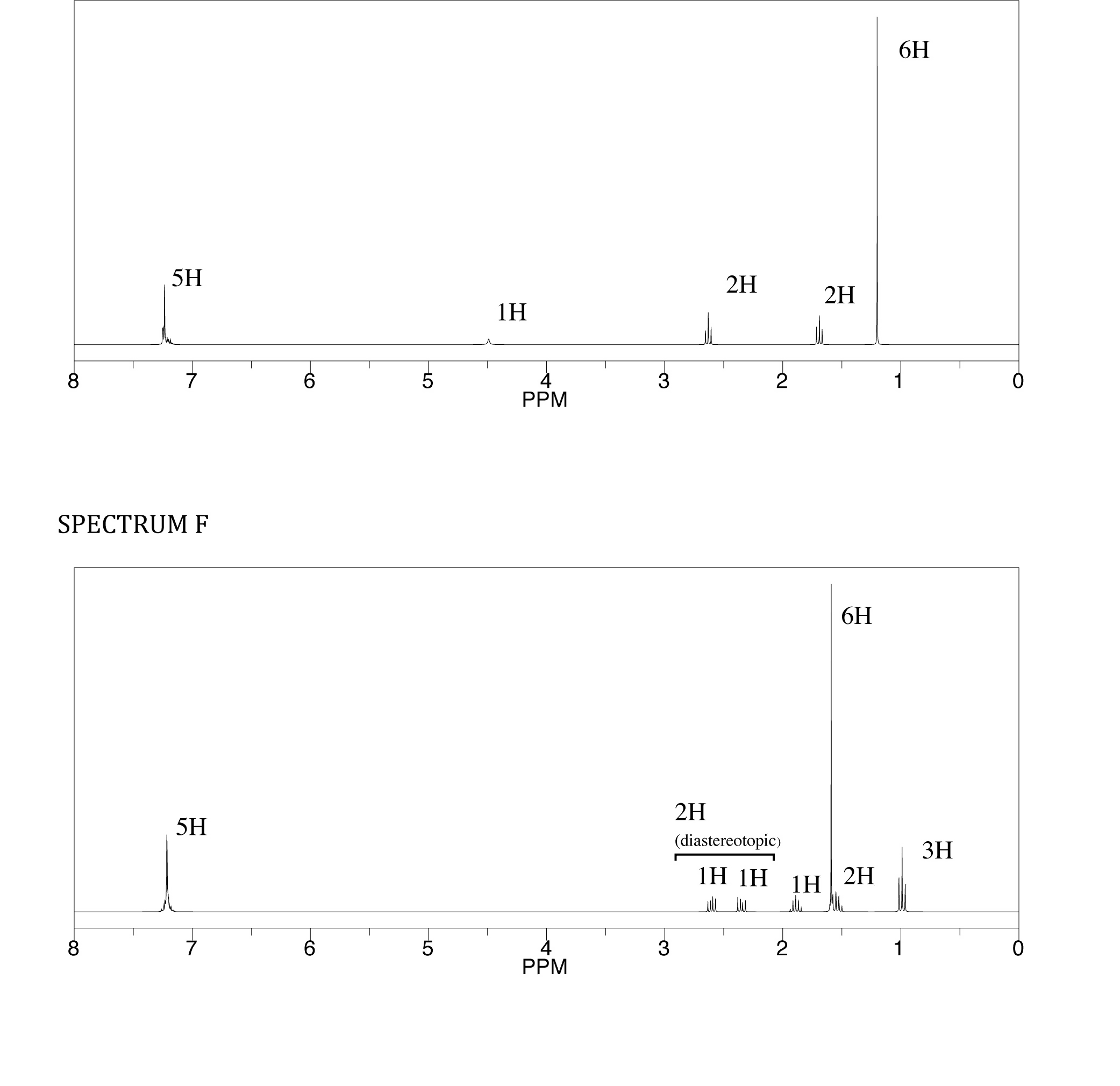
CHM-3120 Final: Unit III and IV Cheat Sheets
60 views3 pages
30 Jun 2017
School
Department
Course
Professor
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related Documents
Related Questions
A process at constant T and P can be described as spontaneous if ÎG < 0 and nonspontaneous if ÎG > 0. Over what range of temperatures is the following process spontaneous? Assume that gases are at a pressure of 1 atm. (Hint: Use the data below to calculate ÎH and ÎS [assumed independent of temperature and equal to ÎH° and ÎS°, respectively] and then use the definition of ÎG.)
| 2 SO3(g) | â | 2 SO2(g) | + | O2(g) | |
| ÎHf° (kJ mol-1) | -395.7 | -296.8 | |||
| S° (J K-1 mol-1) | 256.8 | 248.2 | 205.1 |
ÎH° = kJ
ÎS° = J K-1
The calculated value of T at which ÎG° = 0 is K.
Reaction is spontaneous _________above this temperaturebelow this temperatureat all temperaturesat no temperature.