CHM 11500 Study Guide - Final Guide: Nernst Equation, Electromotive Force, Reduction Potential

27
CHM 11500 Full Course Notes
Verified Note
27 documents
Document Summary
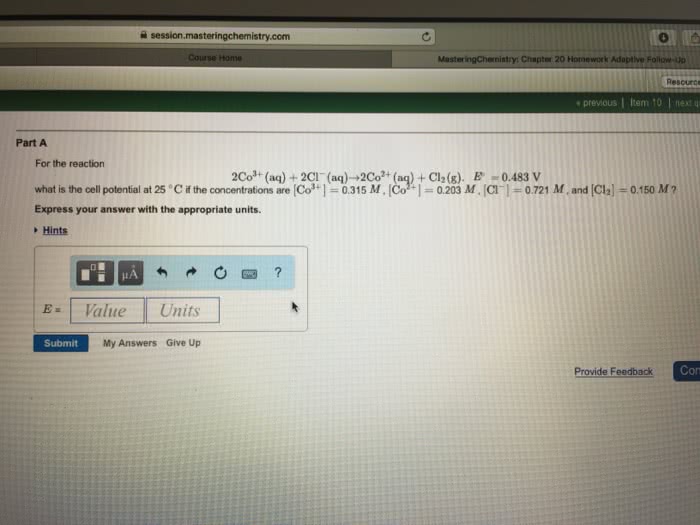
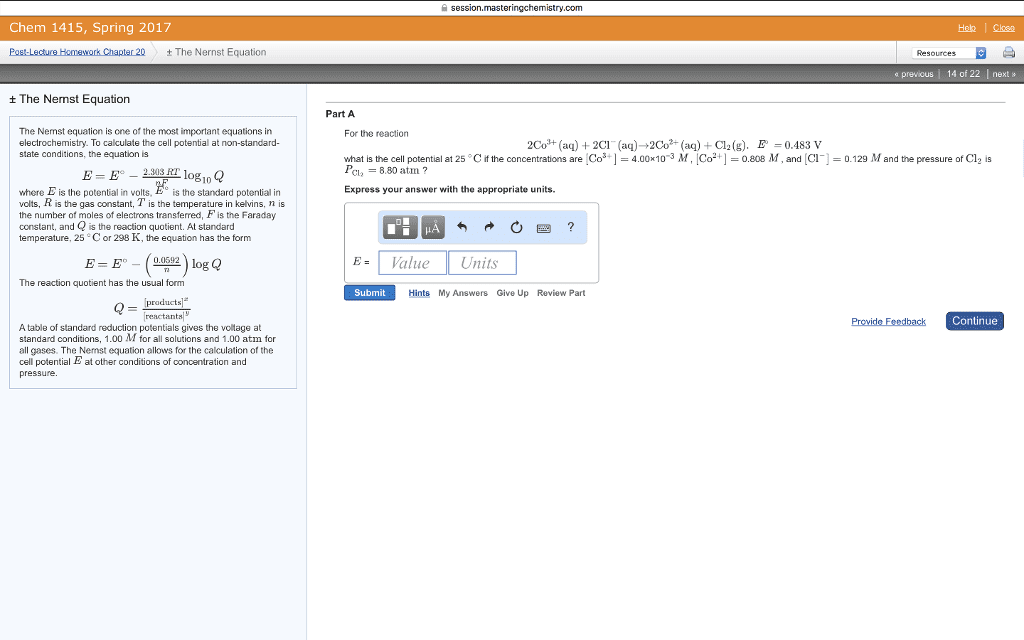
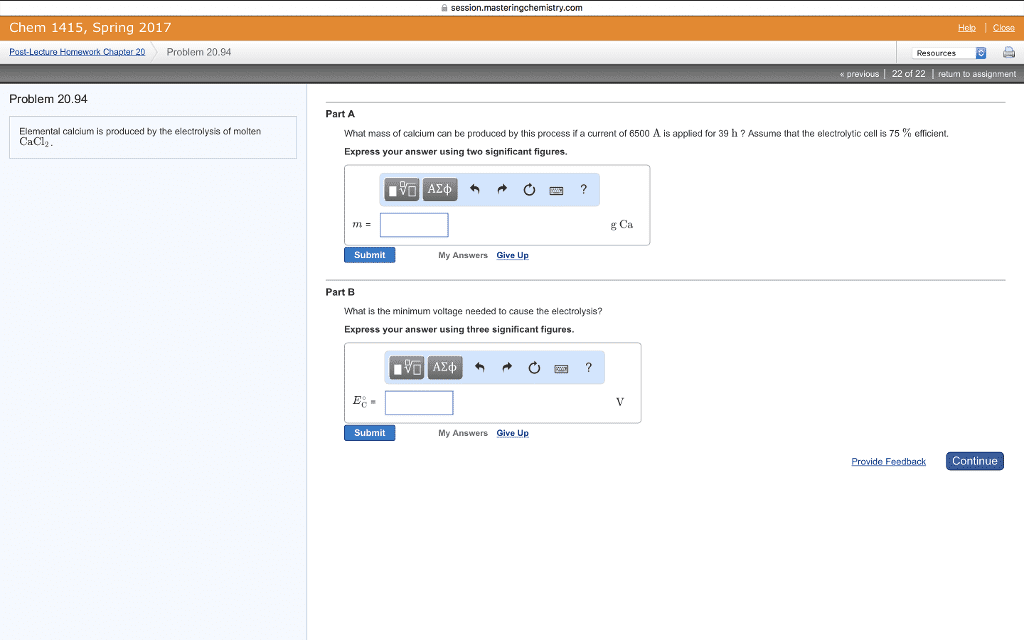
A common term for this difference in electrical potential is electromotive force or emf: needed to solve for the nonstandard cell potential for an electrochemical reaction using. Nernst equation: standard cell potential n (number of electrons in balanced reaction) . Q , reaction coefficient: q to e cell or k, q=1 ecell=ecello, q<1. Ecell< ecello: q=k ecell=0, q0, q>k ecell<0, by definition, if one coulmb of charge passes between 2 electrodes that differ in potential by 1 volt, one joule of energy is realeased. If standard cell potential for a reaction is equal to 0v, go is equal to 0kj. no driving force: g=-nfe constant n=moles electrons exchanged. F=faraday: voltaic cell g<0 system does work on surroundings electrolytic cell . G>0 work is done on the system: electrode at which oxidation occurs is the anode, while electrode where reduction occurs is called cathode, branch of chemistry that deals with the incerconversion of electrical and chemical energy is called electrochemistry.