[BCM 475] - Final Exam Guide - Ultimate 63 pages long Study Guide!
Document Summary
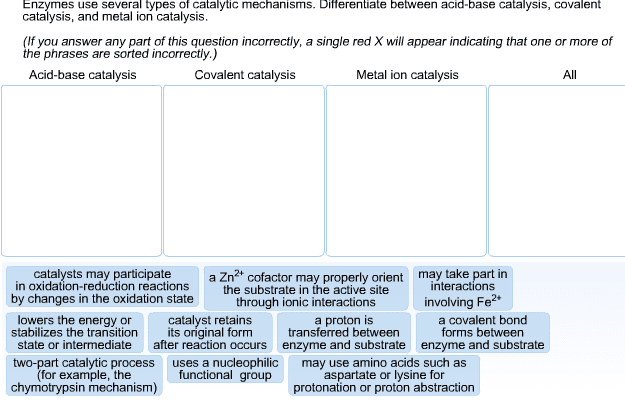
Binding energy free energy released in the formation of a large number of weak interactions between the enzyme and substrate. 2 purposes: establishes substrate specificity | only the correct substrate participates in interactions, increases catalytic efficiency | promote structural changes induced fit. Full complement of interactions occurs in transition state: enzyme-substrate interaction stabilizes ts lowering free energy of activation. Lowers energy or stabilizes the transition state. Covalent catalysis: active site contains reactive group (i. e. , powerful nucleophile functional group) Temporary covalent bond between enzyme and substrate: 2 part process: Breakdown of intermediate: ex: proteolytic enzyme chymotrypsin. General acid-base catalysis: non-water molecule is proton donor/acceptor. Could use amino acids (d aspartate, l) for protonation. Histidine has a pka ~ 7 which allows it to perform as acid/base at physiological ph: ex: chymotrypsin uses h residue as base catalyst. H residue in carbonic anhydrase facilitates removal of h+ from zinc-water molecule. Myosins p of atp is a base to promote hydrolysis.