

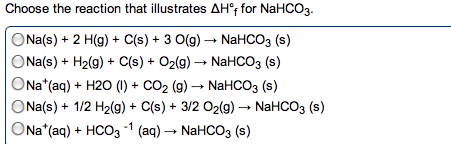






At constant pressure, the combustion of 10.0 g of C2Hg(g) releases 518 kJ of heat. What is AH for the reaction given below? 2 C2H6(g) +- 7 O2(g) rightarrow 4 CO2(g) + 6 H2O(l). -779 kJ -1560 kJ -86.1 kJ -3120 kJ Use the information provided to determine Delta H degree r times n for the following reaction: CH4(g) + 3 Cl2(g) rightarrow CHCI3(I) + 3 HCI(g) Delta H degree r times n = ? -217 kJ -335 kJ +662 kJ +117 kJ -151 kJ Choose the reaction that illustrates Delta H degree f for NaHCO3. Na(s) + 2 H(g) + C(s) + 3 O(g) rightarrow NaHCO3 (s) Na(s) + H2(g) + C(s) + O2(g) rightarrow NaHCO3 (s) Na+(aq) + H2O (I) + CO2 (g) rightarrow NaHCO3 (s) Na(s) + 1/2 H2(g) + C(s) + 3/2 O2(g) rightarrow NaHCO3 (s) Na+(aq) + HCO3 -1 (aq) rightarrow NaHCO3 (s) Use the standard reaction enthalpies given below to determine Delta H degree r times n for the following reaction: 4 SO3(g) rightarrow 4 S(s) + 6 O2(g) Delta H degree r times n = ? Given: SO2(g) rightarrow S(s) + O2(g) Delta H degree r times m = +296.8 kJ 2 SO2(g) + O2(g) rightarow 2 SO3(g) Delta H degree r times n = -197.8 kJ -494.6 kJ -791.4 kJ 1583 kJ -293.0 kJ -692.4 kJ Which of the following statements is TRUE? State functions do not depend on the path taken to arrive at a particular state. Energy is neither created nor destroyed, excluding nuclear reactions. Delta Hr times r can be determined using constant pressure calorimetry. Delta H r times n can be determined using constant volume calorimetry. All of the above are true. How much energy is evolved during the formation of 98.7 g of Fe, according to the reaction below? Fe2O3(s) + 2 Al(s) rightarrow Al2O3(s) + 2 Fe(s) Delta H degree r times n = 852 kJ 01.51 times 103 kJ 241 kJ 0482 kJ 4.20 x 103 kJ 753 kJ Given w = 0, an endothermic reaction has the following. - Delta H and + Delta E Delta H and -Delta E - Delta H and -Delta E + Delta H and +Delta E Which of the following substances (with specific heat capacity provided) would show the greatest temperature change upon absorbing 100.0 J of heat? 10.0 g ethanol, Celharol = 2.42 J/g degree C 10.0 g Ag, CAg = 0.235 J/g degree C 10.0 g Au, CAu = 0.128 J/g degree C 10.0 g Fe, CFe = 0.449 J/g degree C 10.0 g H2O, CHO = 4.18J/g degree C Which of the following processes is endothermic? a hot cup of coffee (system) cools on a countertop the vaporization of rubbing alcohol the chemical reaction in a "hot pack" often used to treat sore muscles the freezing of water the combustion of propane
Show transcribed image text At constant pressure, the combustion of 10.0 g of C2Hg(g) releases 518 kJ of heat. What is AH for the reaction given below? 2 C2H6(g) +- 7 O2(g) rightarrow 4 CO2(g) + 6 H2O(l). -779 kJ -1560 kJ -86.1 kJ -3120 kJ Use the information provided to determine Delta H degree r times n for the following reaction: CH4(g) + 3 Cl2(g) rightarrow CHCI3(I) + 3 HCI(g) Delta H degree r times n = ? -217 kJ -335 kJ +662 kJ +117 kJ -151 kJ Choose the reaction that illustrates Delta H degree f for NaHCO3. Na(s) + 2 H(g) + C(s) + 3 O(g) rightarrow NaHCO3 (s) Na(s) + H2(g) + C(s) + O2(g) rightarrow NaHCO3 (s) Na+(aq) + H2O (I) + CO2 (g) rightarrow NaHCO3 (s) Na(s) + 1/2 H2(g) + C(s) + 3/2 O2(g) rightarrow NaHCO3 (s) Na+(aq) + HCO3 -1 (aq) rightarrow NaHCO3 (s) Use the standard reaction enthalpies given below to determine Delta H degree r times n for the following reaction: 4 SO3(g) rightarrow 4 S(s) + 6 O2(g) Delta H degree r times n = ? Given: SO2(g) rightarrow S(s) + O2(g) Delta H degree r times m = +296.8 kJ 2 SO2(g) + O2(g) rightarow 2 SO3(g) Delta H degree r times n = -197.8 kJ -494.6 kJ -791.4 kJ 1583 kJ -293.0 kJ -692.4 kJ Which of the following statements is TRUE? State functions do not depend on the path taken to arrive at a particular state. Energy is neither created nor destroyed, excluding nuclear reactions. Delta Hr times r can be determined using constant pressure calorimetry. Delta H r times n can be determined using constant volume calorimetry. All of the above are true. How much energy is evolved during the formation of 98.7 g of Fe, according to the reaction below? Fe2O3(s) + 2 Al(s) rightarrow Al2O3(s) + 2 Fe(s) Delta H degree r times n = 852 kJ 01.51 times 103 kJ 241 kJ 0482 kJ 4.20 x 103 kJ 753 kJ Given w = 0, an endothermic reaction has the following. - Delta H and + Delta E Delta H and -Delta E - Delta H and -Delta E + Delta H and +Delta E Which of the following substances (with specific heat capacity provided) would show the greatest temperature change upon absorbing 100.0 J of heat? 10.0 g ethanol, Celharol = 2.42 J/g degree C 10.0 g Ag, CAg = 0.235 J/g degree C 10.0 g Au, CAu = 0.128 J/g degree C 10.0 g Fe, CFe = 0.449 J/g degree C 10.0 g H2O, CHO = 4.18J/g degree C Which of the following processes is endothermic? a hot cup of coffee (system) cools on a countertop the vaporization of rubbing alcohol the chemical reaction in a "hot pack" often used to treat sore muscles the freezing of water the combustion of propane