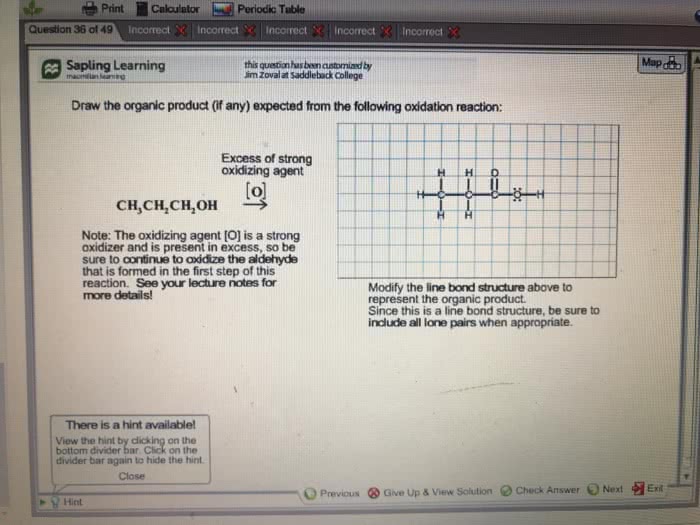
Help with questions 1 and 2 please!
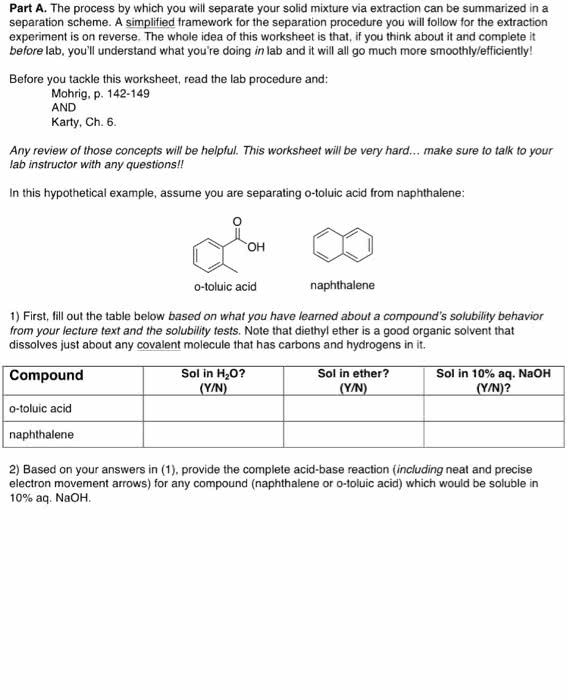
The process by which you will separate your solid mixture via extraction can be summarized in a separation scheme. A simplified framework for the separation procedure you will follow for the extraction experiment is on reverse. The whole idea of this worksheet is that, if you think about it and complete it before lab, you'll understand what you're doing in lab and it will all go much more smoothly/efficiently! Before you tackle this worksheet, read the lab procedure and: Mohrig, p. 142-149 AND Karty, Ch. 6. Any review of those concepts will be helpful. This worksheet will be very hard... make sure to talk to your lab instructor with any questions!! In this hypothetical example, assume you are separating o-toluic acid from naphthalene: 1) First, ill out the table below based on what you have learned about a compound's solubility behavior from your lecture text and the solubility tests. Note that diethyl ether is a good organic solvent that dissolves just about any covalent molecule that has carbons and hydrogens in it. 2) Based on your answers in (1), provide the complete acid-base reaction (including neat and precise electron movement arrows) for any compound (naphthalene or o-toluic acid) which would be soluble in 10% aq. NaOH.