Okay so this is practice for my exam, i have no clue on how toanswer this, please can you guys give me detailed answers to thesequestions so i can have a base to study from. Highest pointsrewarded to the person being the most understandable andspecifc
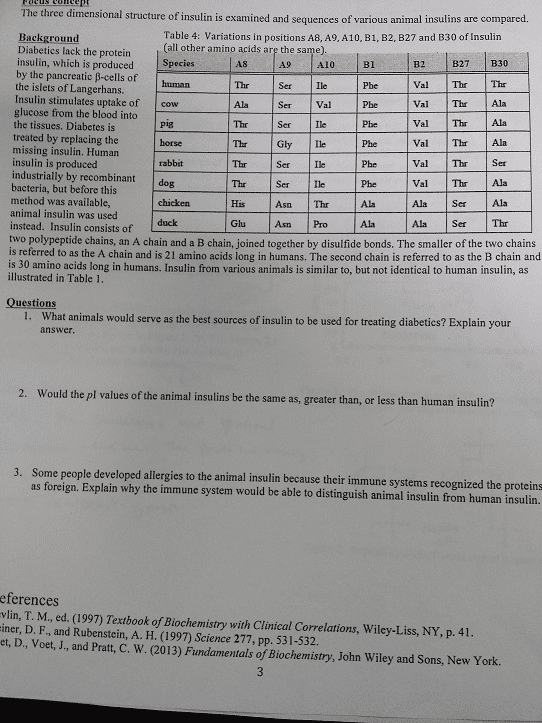
The three dimensional structure of insulin is examined and sequences of various animal insulins are compared. Table 4: Variations In positions A8, A9, A10, Bl. B2, B27 and B30 of Insulin (all other amino acids are the sample). Diabetics lack the protein insulin, which is produced by the pancreatic beta-cells of the islets of Langerhans. Insulin stimulates uptake of glucose from the blood into the tissues. Diabetes is treated by replacing the missing insulin. Human insulin is produced industrially by recombinant bacteria, but before this method was available, animal insulin was used instead. Insulin consists of two polypeptide chains, an A chain and a B chain, joined together by disulfide bonds. The smaller of the two chains is referred to as the A chain and is 21 amino acids long in humans. The second chain is referred to as the B chain and is 30 amino acids long in humans. Insulin from various animals is similar to, but not identical to human insulin, as illustrated in Table 1. What animals would serve as the best sources of insulin to be used for treating diabetics ? Explain your answer. Would the pI values of the animal insulins be the same as, greater than, or less than human insulin ? Some people developed allergies to the animal insulin because their immune systems recognized the proteins as foreign. Explain why the immune system would be able to distinguish animal insulin from human insulin. References T. M., ed. (1997) Textbook of Biochemistry with Clinical Correlations, Wiley-Liss, NY, p. 41. D. F., and Rubenstein, A. H. (1997) Science 277, pp. 531-532. D., Voet, J., and Pratt, C. W. (2013) Fundamentals of Biochemistry, John Wiley and Sons, New York.
Show transcribed image text The three dimensional structure of insulin is examined and sequences of various animal insulins are compared. Table 4: Variations In positions A8, A9, A10, Bl. B2, B27 and B30 of Insulin (all other amino acids are the sample). Diabetics lack the protein insulin, which is produced by the pancreatic beta-cells of the islets of Langerhans. Insulin stimulates uptake of glucose from the blood into the tissues. Diabetes is treated by replacing the missing insulin. Human insulin is produced industrially by recombinant bacteria, but before this method was available, animal insulin was used instead. Insulin consists of two polypeptide chains, an A chain and a B chain, joined together by disulfide bonds. The smaller of the two chains is referred to as the A chain and is 21 amino acids long in humans. The second chain is referred to as the B chain and is 30 amino acids long in humans. Insulin from various animals is similar to, but not identical to human insulin, as illustrated in Table 1. What animals would serve as the best sources of insulin to be used for treating diabetics ? Explain your answer. Would the pI values of the animal insulins be the same as, greater than, or less than human insulin ? Some people developed allergies to the animal insulin because their immune systems recognized the proteins as foreign. Explain why the immune system would be able to distinguish animal insulin from human insulin. References T. M., ed. (1997) Textbook of Biochemistry with Clinical Correlations, Wiley-Liss, NY, p. 41. D. F., and Rubenstein, A. H. (1997) Science 277, pp. 531-532. D., Voet, J., and Pratt, C. W. (2013) Fundamentals of Biochemistry, John Wiley and Sons, New York.


