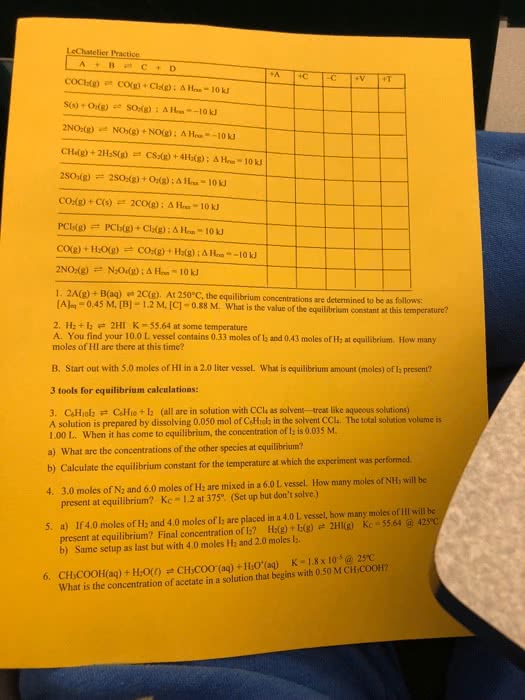
LeChatelier Practice A+B C+D 1. 2A(g) + B(ag) et 2C(g). At 250°C, the equilibrium concentrations are determined to be as follonws: AJo-0.45 M, [B] -12 M, IC]-0.88 M. What is the value of the equilibrium constant at this temperat 2. H2+ 12 2HI K-55.64 at some temperature A. You find your 10.0 L vessel contains 0.33 moles of l2 and 0.43 moles of H at equilabrium. How many moles of HI are there at this time? B. Start out with 5.0 moles of HI in a 2.0 liter vessel. What is equilibrium amount (moles) of Ia present? 3 tools for equilibrium calculations: 3. CsHiolzCsHio +l2 (all are in solution with CCla as solvent-treat like aqueous solutions) A solution is prepared by dissolving 0.050 mol of C&Hioly in the solvent CCl. The total solution volume is 1.00 L. When it has come to equilibrium, the concentration of la is 0.035 M. a) What are the concentrations of the other species at eoquilibrium? b) Calculate the equilibrium constant for the temperature at which the experiment was performed. 4. 3.0 moles of N2 and 6.0 moles of H2 are mixed in a 6.0L vessel. How many moles of NHs will be present at equilibrium? Kc-1.2 at 375 (Set up but don't solve.) If4.0 moles of H2 and 4.0 moles of l2 are placed in a 4.0 L vessel, how many moles of Hl will be present at equilibrium? Final concentration of Iz? Hag) + L() b) Same setup as last but with 4.0 moles H2 and 2.0 moles l2. 5. a) 2H!(g) Kc = 55.64 @ 425â K-1.8 x 10am CHyC00(aq) + HO'(aq) CHCOOH(aq) + H20(1) What is the concen 6. tration of acetate in a solution that begins with 0.50 M CH,COOH?