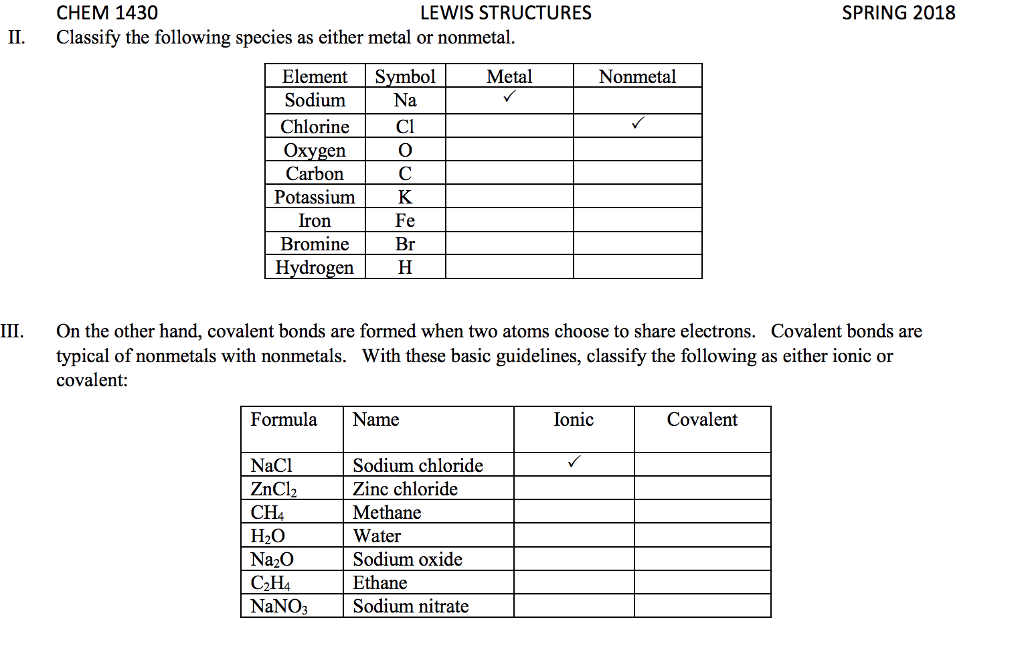
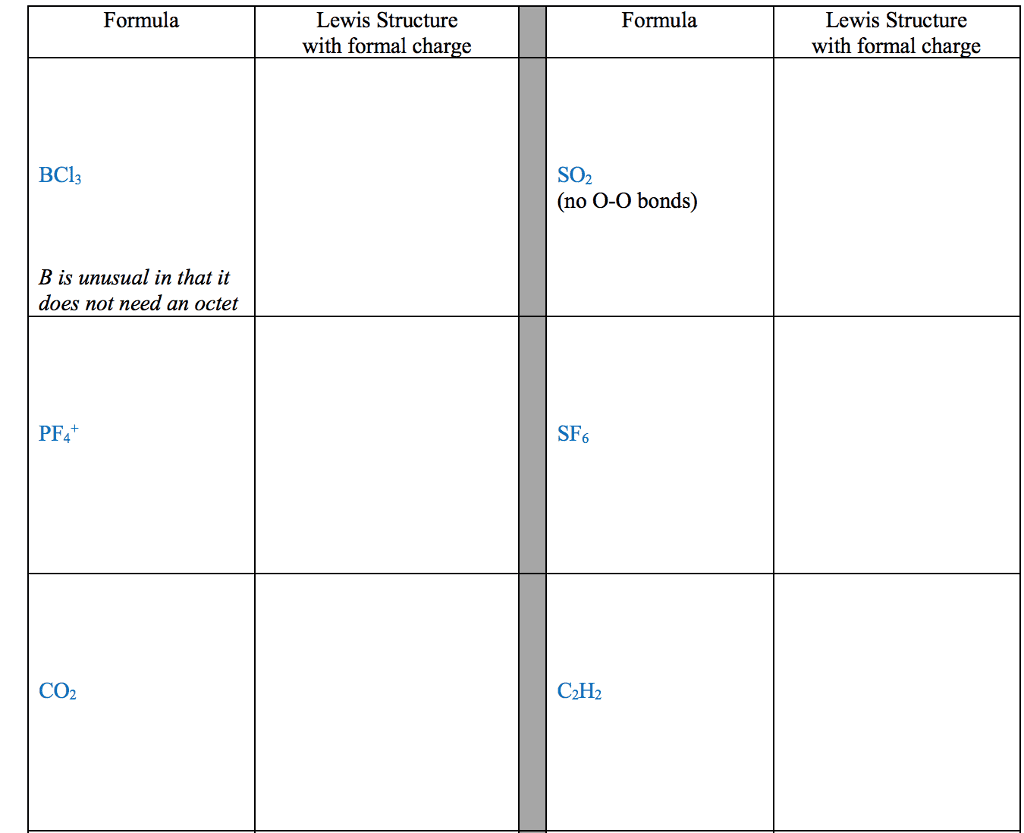
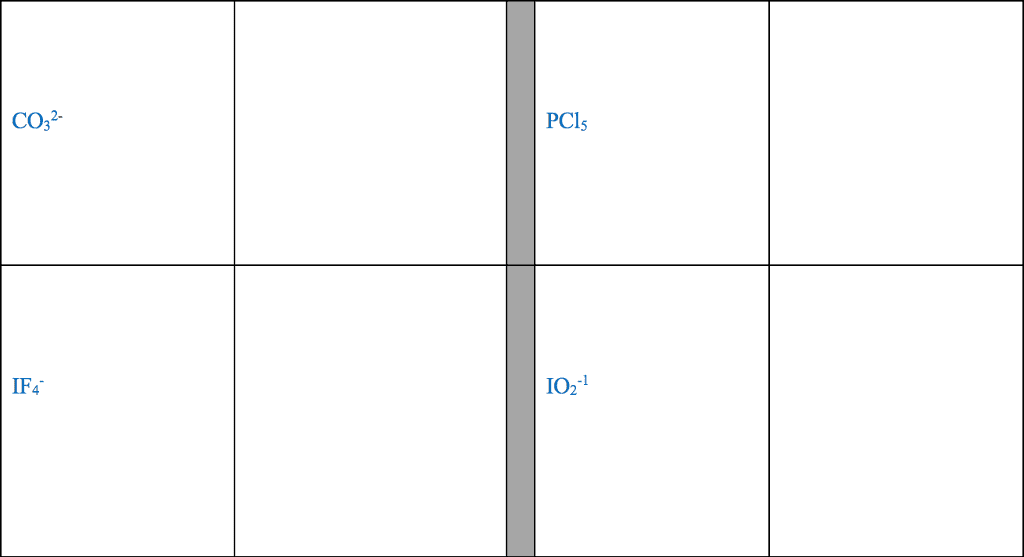
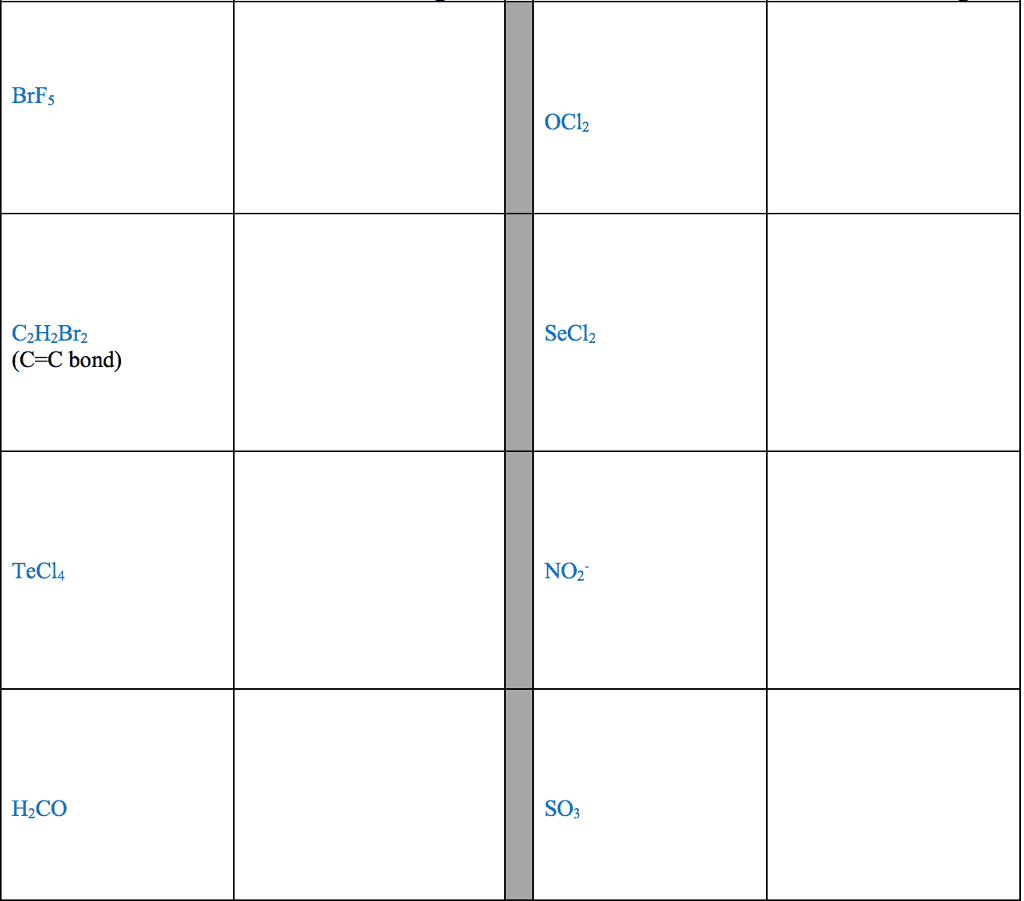
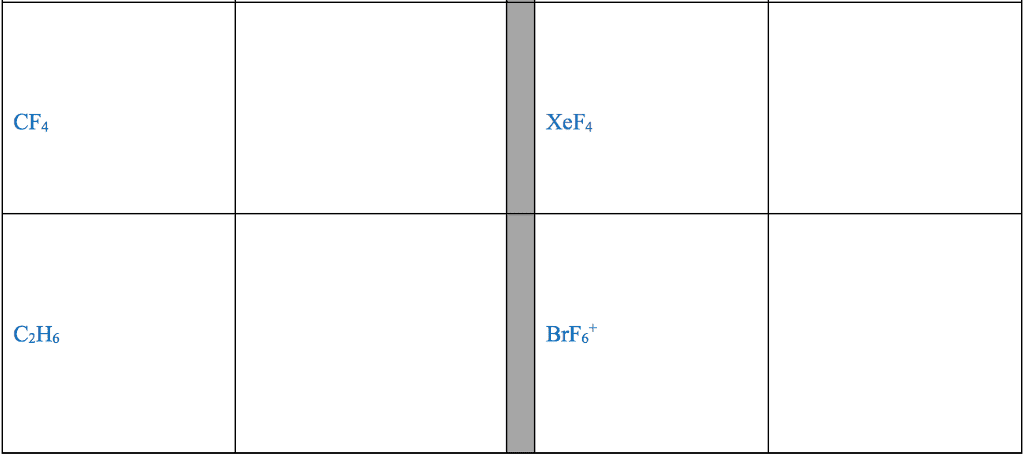
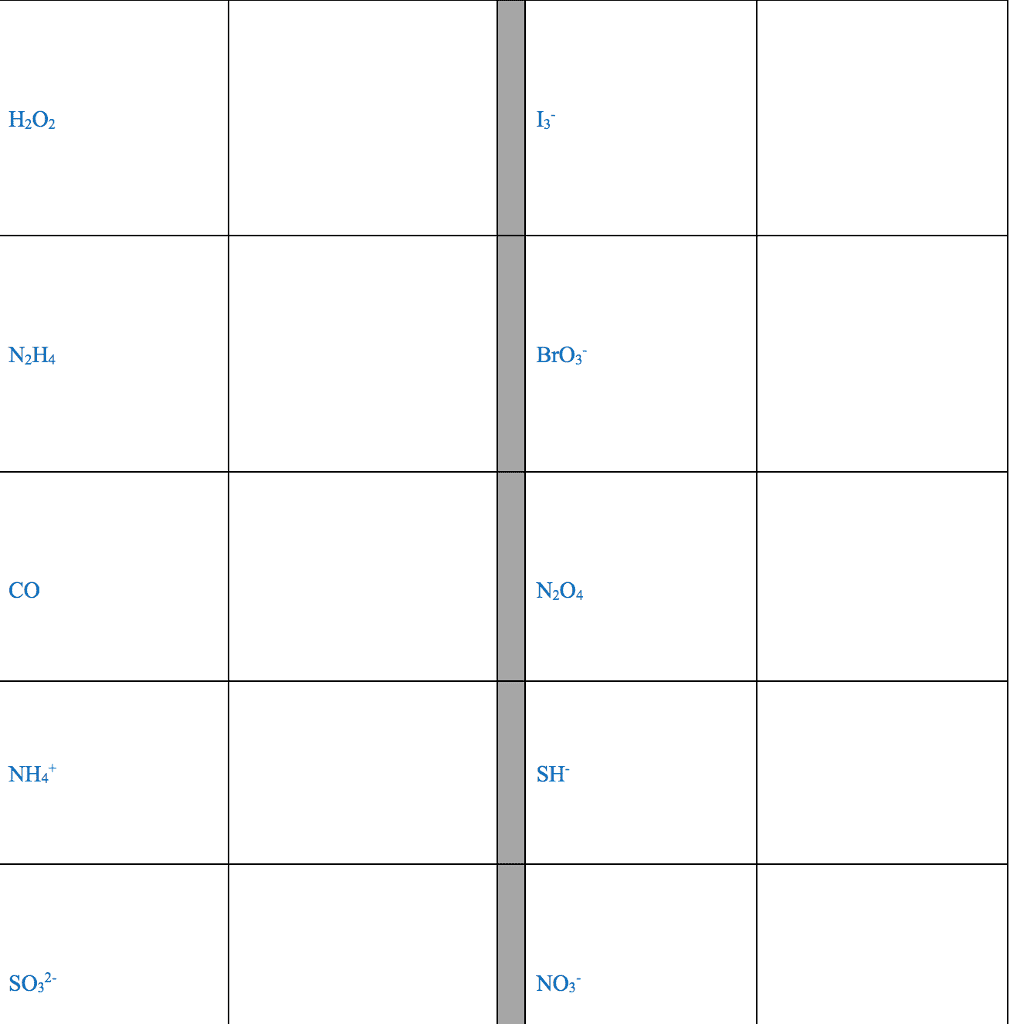
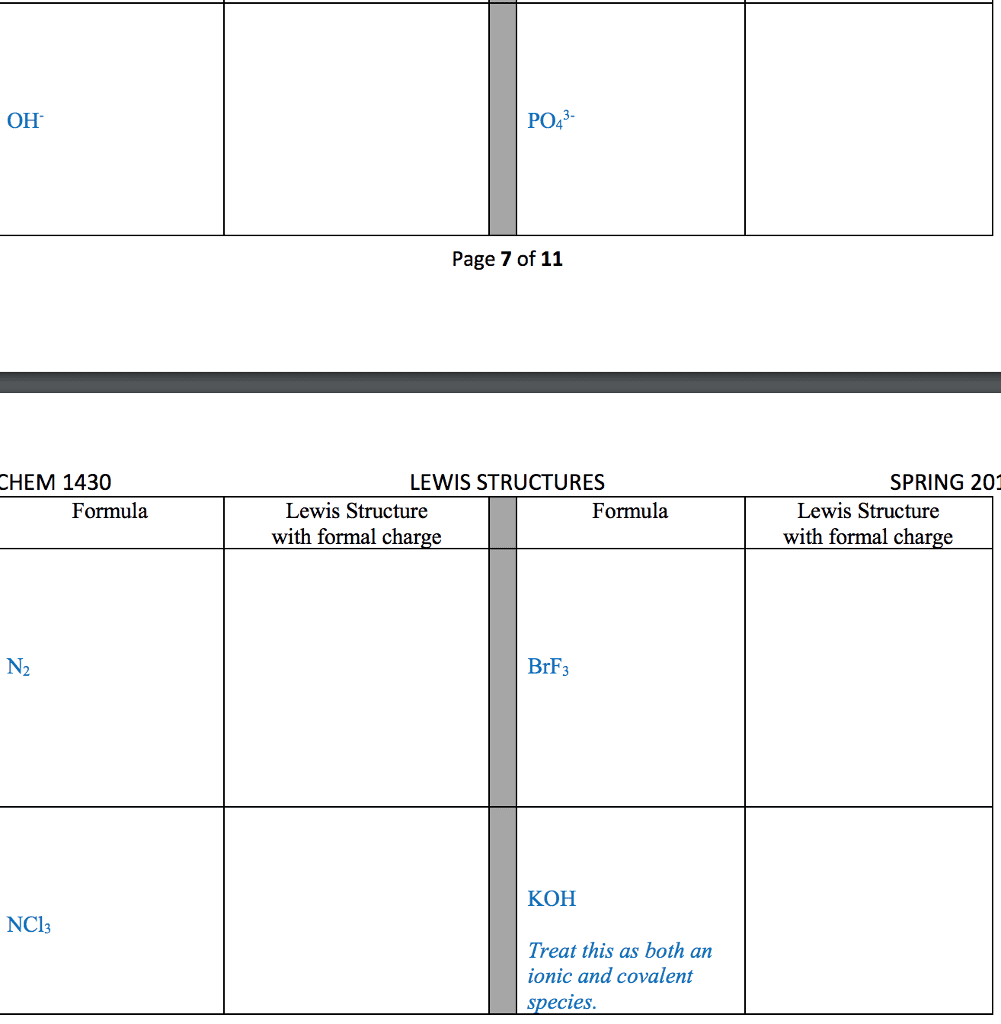
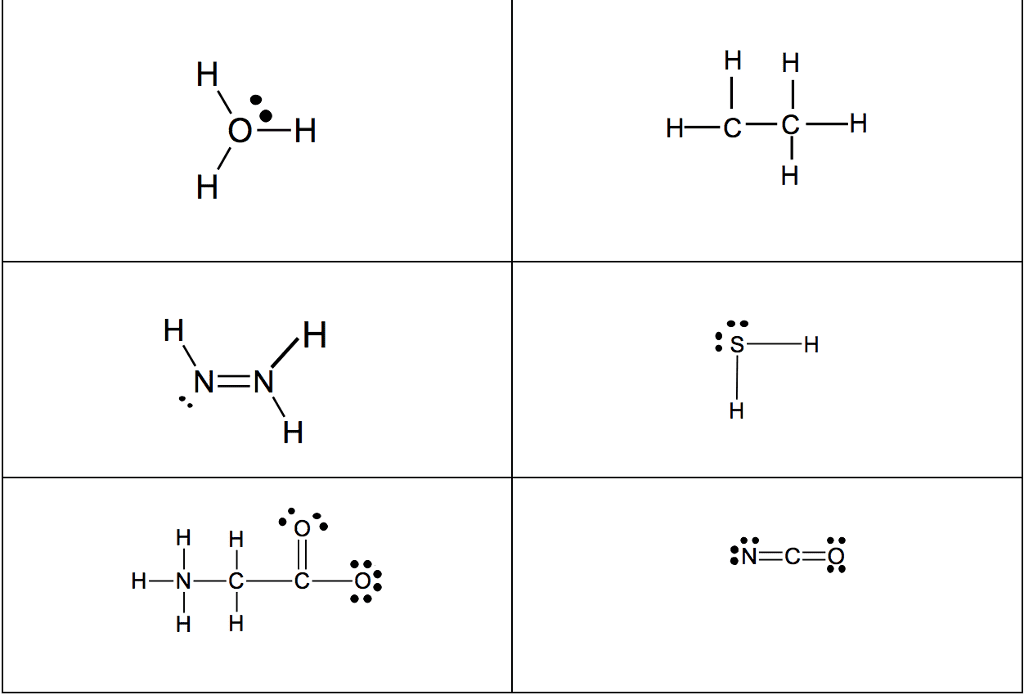
STRC 434: Wricing Lewis Symbols and Lewis Strucnures (4) Write the Lewis structure for the ionic com 5. Construct the Lewis structure model for the cova pound potassium chioride in the box. lent compound sulfur difluoride (SF2) using the tollow ing steps (1) atom is The number of valence electrons in the sulfur . The total number of valence electrons is the total Lewis structure of potassium chloride How does the written Lewis structure for po, in the two fluorine atoms isHence number ol valence electrons in SF's (5) tassium chioride differ from that ot hydrogen chloride? (2) Arrange the atomic cores for the two fluorine atoms with the sultur atom as the central atom in the space below. (3) Rewrite the atomic cores in the space below. Place an electron pair between each fluorine and sulfur core. Write the Lewis structure for SF2 in the box, lent compound hydrogen sulfide (H2S) using the fol- distributing the remaining valence electrons so that all 4. Construct the Lewis structure model for the cova- (4) lowing steps. three atoms are in accordance with the octet rule. (1) The total number of valence electrons in the two hydrogen atoms is ; the sulfur atom has Hence, the total number of valence electrons in the compound is Lewis structure of SF2 (2) Write the atomic cores for the hydrogen atoms and the sulfur atom in the space below. Because sulfur is the central atom in this molecule, ar- range the cores so that the hydrogen atoms are on ei ther side of the sulfur atom. 6. Construct the Lewis structure model for the cova- lent compound methylene chloride (CH2C) The number of valence electrons in the car- The total number of valence elec (1) bon atom is trons in the two hydrogen atoms is number of valence electrons in the two chlorine atoms is trons in CH2C12 is The total (3) Rewrite the atomic cores in the space below. Place an electron pair between each hydrogen atom and the suilfur atom. Hence, the total number of valence elec- (2) Position the atomic core for carbon as the (4) Write the Lewis structure for hydrogen sulfide central atom in the space below, and place the other in the box, distributing the remaining valence electrons cores around it so that the hydrogen atoms are in accordance with the duet rule and the sulfur atom is in accordance with the octet rule. (3) Rewrite your arrangement of atomic cores ir the space below, joining the cores with electron pairs to form the necessary bonds. Lewis structure of H2S