BIOCHEM 524 Study Guide - Quiz Guide: Non-Covalent Interactions, Relative Permittivity, Hydrogen Bond
Document Summary
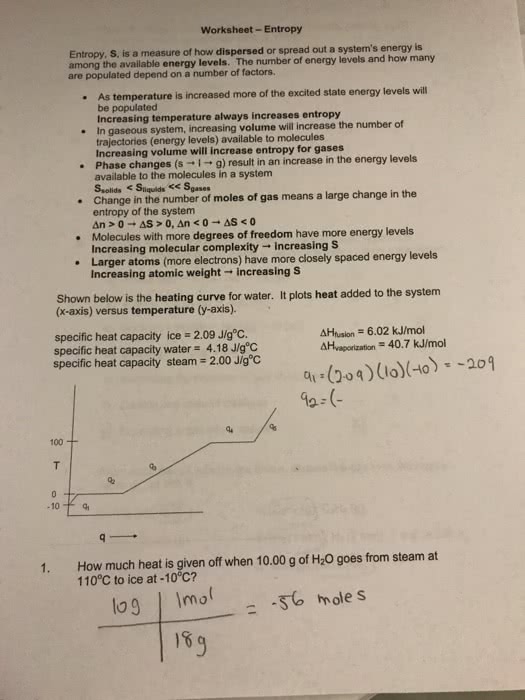
Here are some very brief guidelines as to what i would like you to know for quiz 1. And also archaea: organisms that inhabit exteme environments, but are found most everywhere: eukaryotes: usually larger than prokaryotic cells and contain a nucleus and membrane-bound cellular compartments. H bonds: second strongest noncovalent bond, forms between h attached to o, s, or n and forms between open o, s, or n. Chapter 3: experimental basis for model of dna double helix (in a very general sense) Chargaff"s rules: a & t and g & c are present in the same concentrations in dna. In dna bases stack 3. 4a apart: properties of dna double helix single turn is 34a (10 bp, the entire helix is 20a wide, there is a major and minor groove. Waals contact the side chains extend out from the helix.