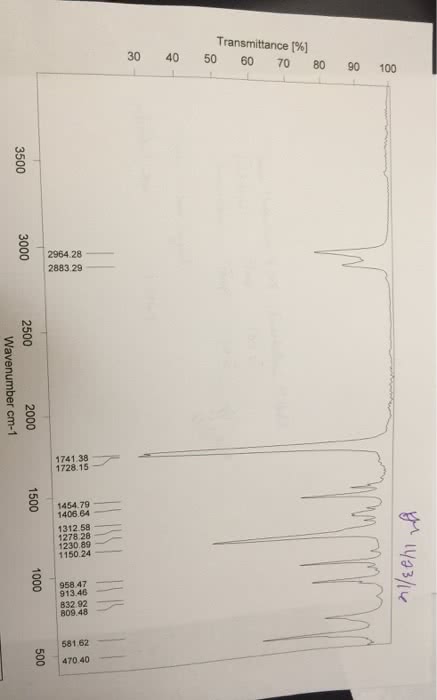
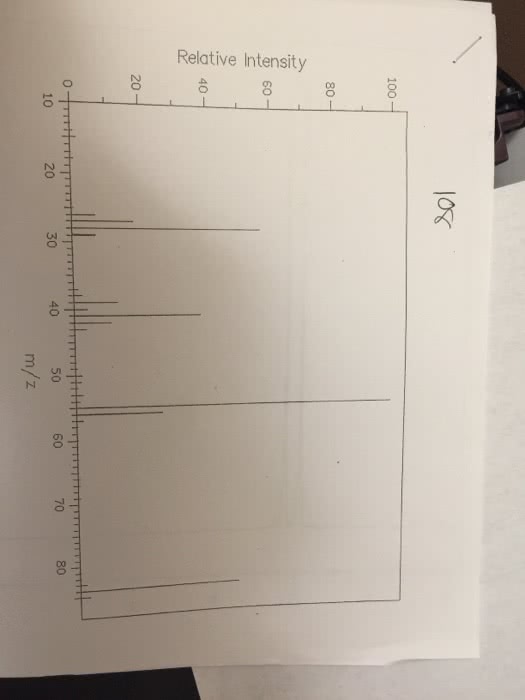
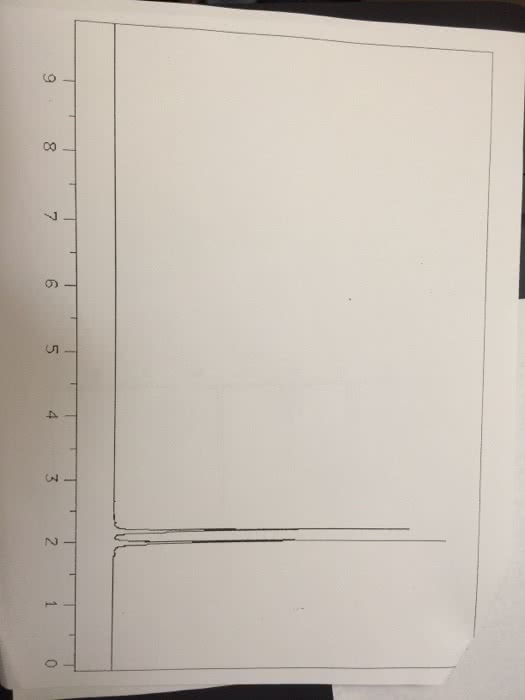
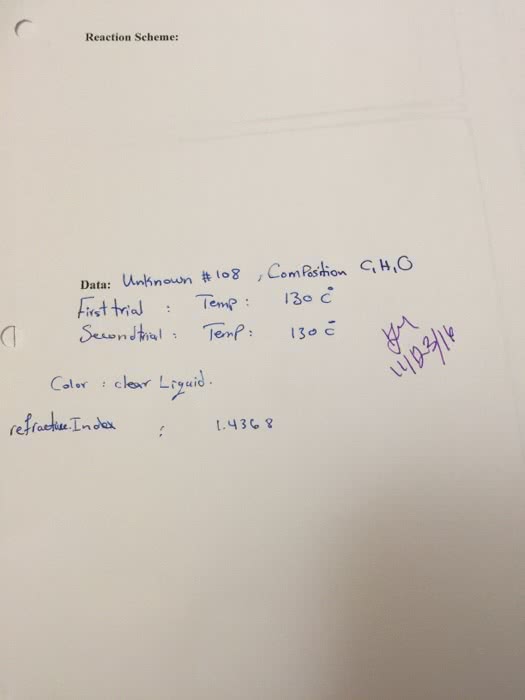CHEM 112 : Lab Report 3.docx
Get access

Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
Just need help with a few questions from my lab!
Lab Notes:
Experiment 1:
Tube 1: No bubbles produced
Tube 2: Bubbles produced, slowly and sporadically
Tube 3: Bubbles produced, quickly and constantly
Tube 4: Bubbles produced, slowly and sporadically
Tube 5: No bubbles produced
Optimum temp for catalase activity: 40 C
Experiment 2:
Tube 1: 4 min, 42.38 sec
Tube 2: 5 min, 4.37 sec
Tube 3: 5 min, 18.26 sec
Tube 4: 5 min, 28.36 sec
Concentration: 10 mL 3% hydrogen peroxide
Experiment 3:
Tube 1: No bubbles produced
Tube 2: Bubbles produced, quickly
Tube 3: No bubbles produced
Optimum pH for catalase activity: pH 6 buffer
Lab Questions
Experiment 1:
1. Why did catalase fail to function at 80 °C?
2. What caused the bubbles to form when you added the catalase to the hydrogen peroxide and water mixture at 40 °C?
A. Catalase activity heated the solution to its boiling point.
B. Hydrogen gas formed during the formation of hydrogen peroxide.
C. Oxygen gas formed during the decomposition of hydrogen peroxide.
D. The constant temperature bath heated the solution to its boiling point.
Experiment 2
3. Before the catalase concentration was saturated with hydrogen peroxide, why did the enzyme function vary with substrate concentration?
4. Why did the catalase function level off when different hydrogen peroxide concentrations were tested?
Experiment 3
5. Why did catalase only function in a limited range of pH values?
6. Based on your results from this lab, would you expect the hydrogen peroxide you buy from the store to bubble like soda when you open it?
| A. Yes, hydrogen peroxide is only stable in the presence of catalase. Without it, store bought hydrogen peroxide decomposes quickly. | ||||||||||||||
| B. No, catalase sped up the rate of the reaction. Store bought hydrogen peroxide will decompose into water and oxygen gas much too slowly for visible bubbles to form. | ||||||||||||||
| C .No, hydrogen peroxide can only decompose into water and oxygen gas in the presence of catalase. | ||||||||||||||
| D. Yes, catalase had little effect on hydrogen peroxide decomposition. It would bubble quickly no matter what. 7. Do the optimal conditions for catalase functioning that you found correspond with the conditions in the mammalian liver where it came from?
|