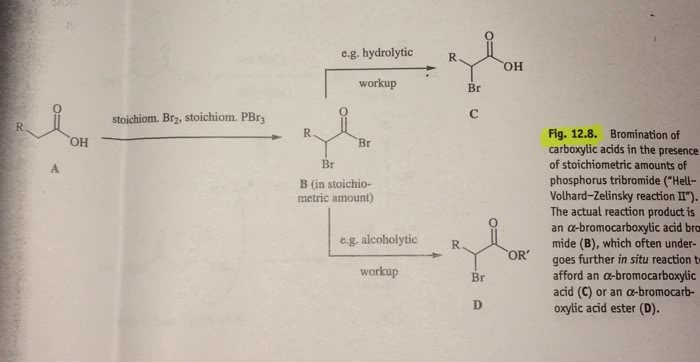
Starting from any hydrocarbon (using the reactions provided) present a reasonable synthetic scheme (Scheme 1) for the synthesis of ISOBUTYL FORMATE (C5H10O2). Please draw out the scheme starting from any hydrocarbon and end the scheme with isobutyl formate as the final product.
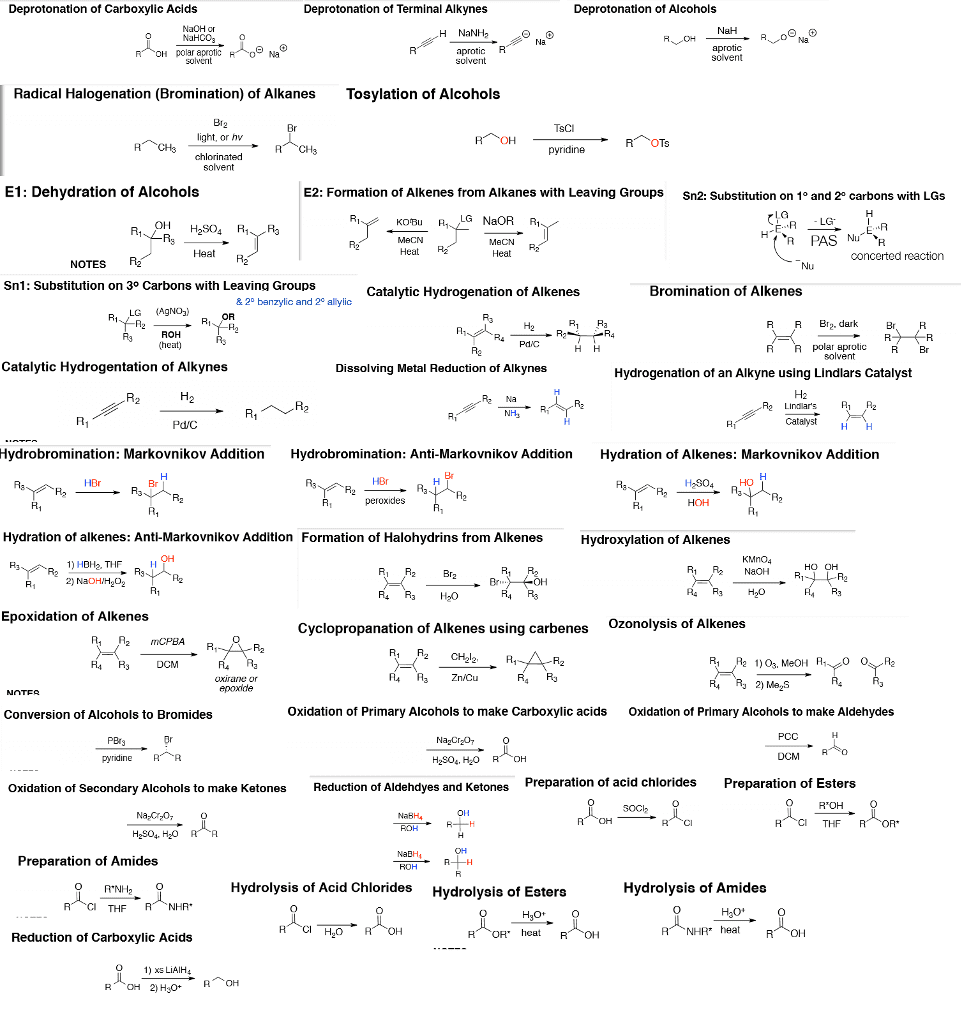
Deprotonation of Carboxylic Acids Deprotonation of Terminal Alkynes Deprotonation of Alcohols Na0H or NaHCO30 H NaNH OH polar aprotic oeNaO aprotic R Radical Halogenation (Bromination) of Alkanes Tosylation of Alcohols TsCI light, or hv pyridine E1: Dehydration of Alcohols E2: Formation of Alkenes from Alkanes with Leaving Groups Sn2: Substitution on 1° and 2 carbons with LGs LG NaOR R1 LG OH Heat Heat R2 Heat R concerted reaction NOTES R2 Sn1: Substitution on 3° Carbons with Leaving Groups Catalytic Hydrogenation of Alkenes Bromination of Alkenes & 20 banzylic and 2c allylic LG (AgNO3ã R1 a R R B.dark ROH heat R R polar eprotic Br Catalytic Hydrogentation of Alkynes Dissolving Metal Reduction of Alkynes Hydrogenation of an Alkyne using Lindlars Catalyst H2 R2 LindiasP2 Catalyst H H Hydrobromination: Markovnikov Additio Hydrobromination: Anti-Markovnikov Addition Hydration of Alkenes: Markovnikov Addition peroxides Hydration of alkenes: Anti-Markovnikov Addition Formation of Halohydrins from Alkenes Hydroxylation of Alkenes 1) HBH2, THF H KMno4 Rı R2 NaOH HO OH Epoxidation of Alkenes Cyclopropanation of Alkenes using carbenes Ozonolysis of Alkenes A2 mCPBA R4 DCM R R3Zn/Cu R4 Ra UXANEN or Conversion of Alcohols to Bromides Oxidation of Primary Alcohols to make Carboxylic acids Oxidation of Primary Alcohols to make Aldehydes pyridine R︿R H2S0, H20 Oxidation of Secondary Alcohols to make Ketones Reduction of Aldehdyes and Ketones Preparation of acid chlorides Preparation of Esters O R'OH NaC0 H2S04 H2O NaBH Preparation of Amides ROH Hydrolysis of Acid Chlorides Hydrolysis of Estera Hydrolysis of Amides H3OO heat R NHR, heat R︿OH Reduction of Carboxylic Acids 0 1)xs LiAlH4