CH 301 Midterm: thermodynamics study guide
50 views3 pages
Document Summary
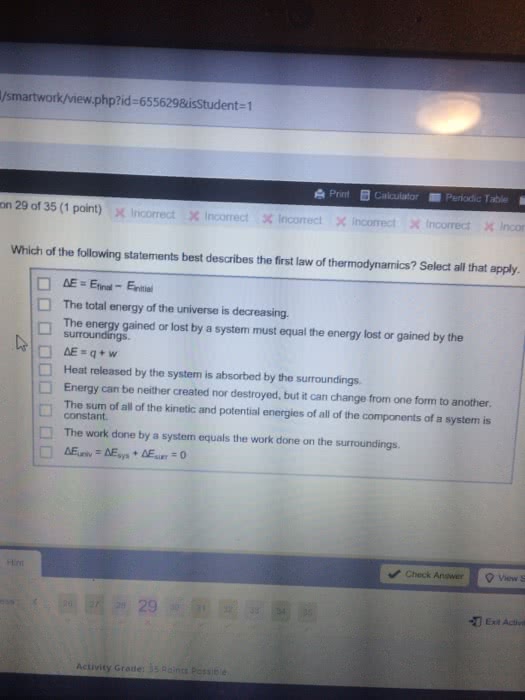
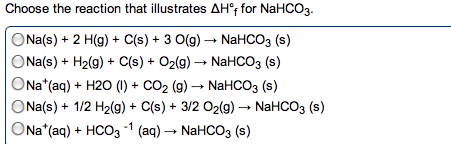
Th er mo dy na mi cs test review state functions are properties that are path dependent. Extensive properties change with amount of substance. Intensive properties do not change with amount of substance first law of thermodynamics states that energy is neither created nor destroyed, only changed in form. Where w can equal -p v (1 latm = 101. 325 j) or - nrt. +w: surroundings do work on the system. +q: absorbed heat equipartition theorem: each particle contributes to u by kt for each mode. (3)rt enthalpy: heat flow at constant pressure between systems and surroundings. H vap is huge because you have to break all imfs to vaporize. Standard conditions are 1 molar concentration, 298k, 1 atm. Bond energies: amount of energy required to break 1 mole of bond. Q = nrtln(v 2 /v 1 ) calorimetry: measuring q output. To convert: u = h - nrt.