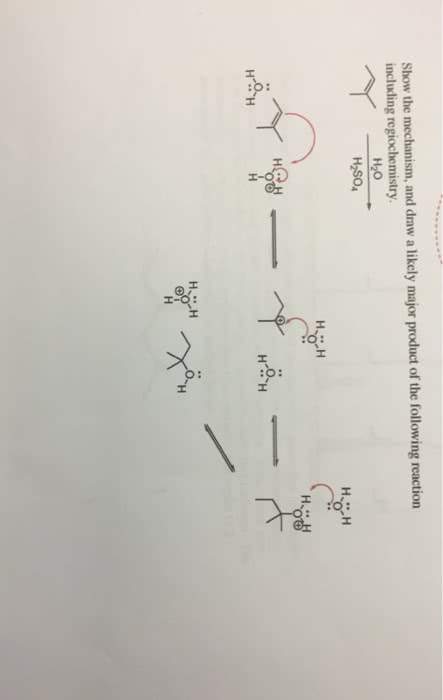
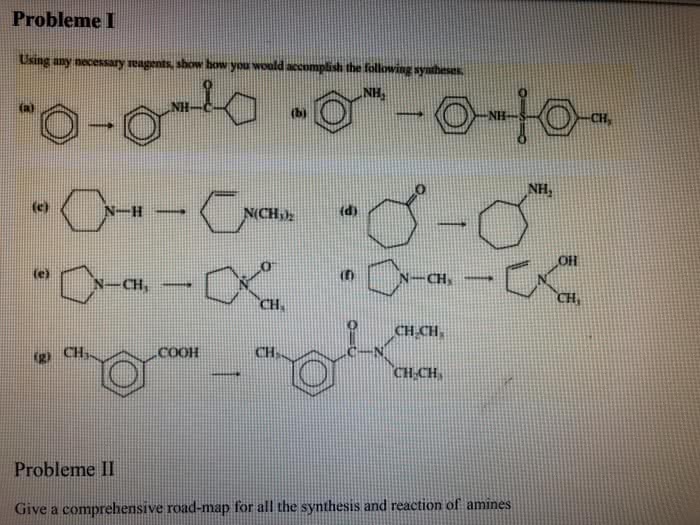
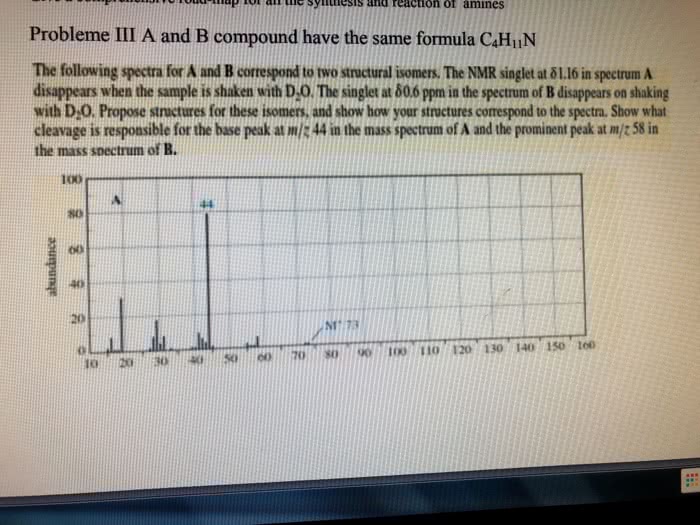
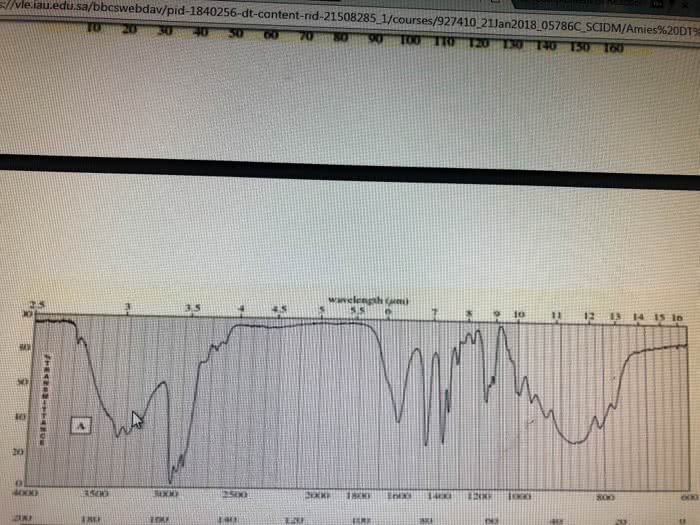
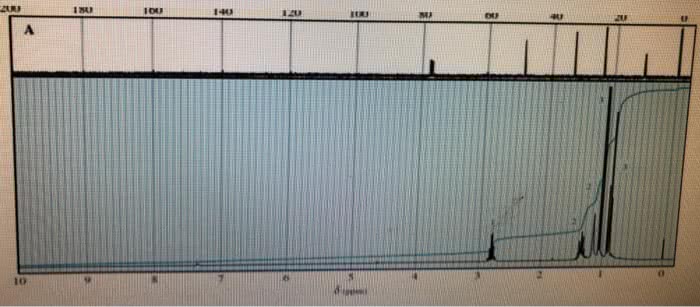
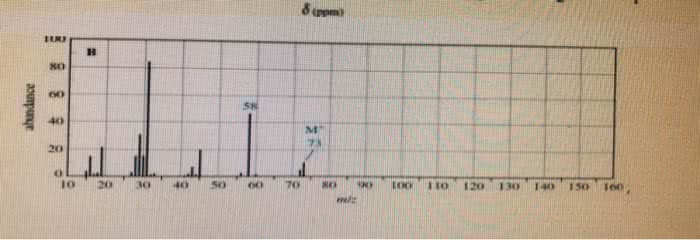
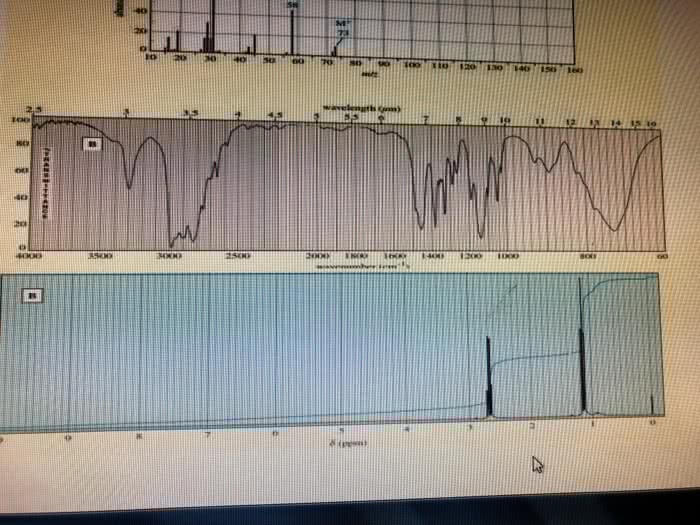
CHE 112 MIDTERM EXAM PRACTICE QUESTIONS 4. The characteristically pungent odor of'skunk spray' is due to the presence of sulfur containing compounds called mercaptans. At 28.0°C, the vapor pressure ofone of these compounds, n. mercaptan, is 175 torr. The molar mass of the mercaptan is 75.16 g/mol and it's density is 0.84 g/mL. prop What is the vapor pressure, at 28.0 â, of a solution made by mixing 100.0 mL of n-pro mercaptan with 0.250 mol of a nonvolatile nonelectrolyte solute? (6 pts) a. b. What is the vapor pressure, at 28.0 °C, of a solution made by mixing 100.0 mL of n-prop mercaptan and 100.0 g CH,CN P-102 torr), if Raoult's Law is obeyed? (6 pts) Which solution of mercaptan above will snell more strongly of the skunk odor, the solution e.