CHEM 243 Chapter 9.1: 3-Scan from a McGill uPrint device (1)
Get access


Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
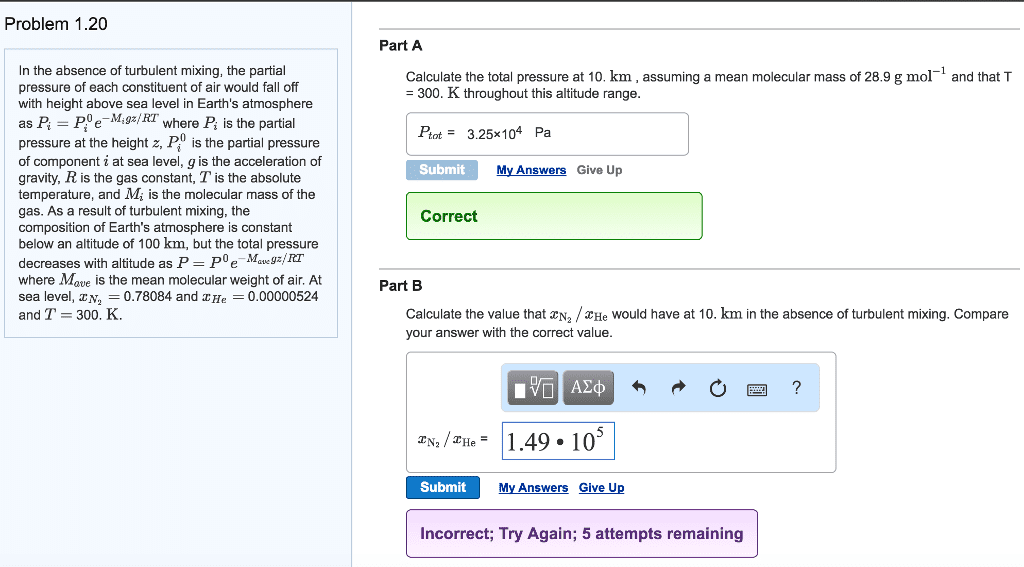
| Problem 1.20 In the absence of turbulent mixing, the partial pressure of eachconstituent of air would fall off with height above sea level inEarth's atmosphere asPi=P0ie?Migz/RTwhere Pi is the partial pressure at the heightz, P0i is the partial pressure ofcomponent i at sea level, g is the accelerationof gravity, R is the gas constant, T is theabsolute temperature, and Mi is the molecularmass of the gas. As a result of turbulent mixing, the compositionof Earth's atmosphere is constant below an altitude of 100 km, butthe total pressure decreases with altitude asP=P0e?Mavegz/RTwhereMave is the mean molecularweight of air. At sea level, xN2= 0.78084 andxHe=0.00000524 and T= 300.K. | Part A Calculate the total pressure at 8.0km , assuming a meanmolecular mass of 28.9 g mol?1 and that T = 300. K throughout thisaltitude range.
SubmitMy AnswersGiveUp Part B Calculate the value that xN2/xHe would have at8.0km in the absence of turbulent mixing. Compare your answer withthe correct value.
SubmitMy AnswersGiveUp |