CHEM 1A03 Chapter Notes -Ionic Bonding, Fluorine, Electronegativity

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
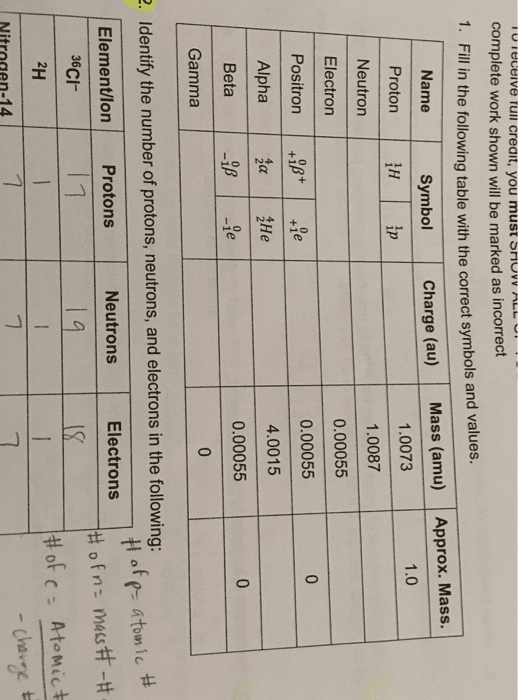
A is the mass number which is the total number of protons and neutrons. Z is the atomic number- the number of protons and electrons in each atom of the element. Isotopes: the same atomic number but different atomic masses due to different numbers of neutrons. Radioisotope: an isotope of a chemical element that has an unstable nucleus and emits radiation during its decay to a stable form, have important uses in medical diagnosis, treatment, and research. Half-life: the length of time it takes for a substance undergoing decay to decrease by half. The volume of space around the nucleus where electrons are most likely found is called the orbital. Those electrons available for bonding are called valence electrons. The number and arrangement of its valence electrons in these s and p orbitals determine the behaviour of the atom. Ionic bond: complete transfer of electron(s) covalent bond: electron(s) shared in orbital space.