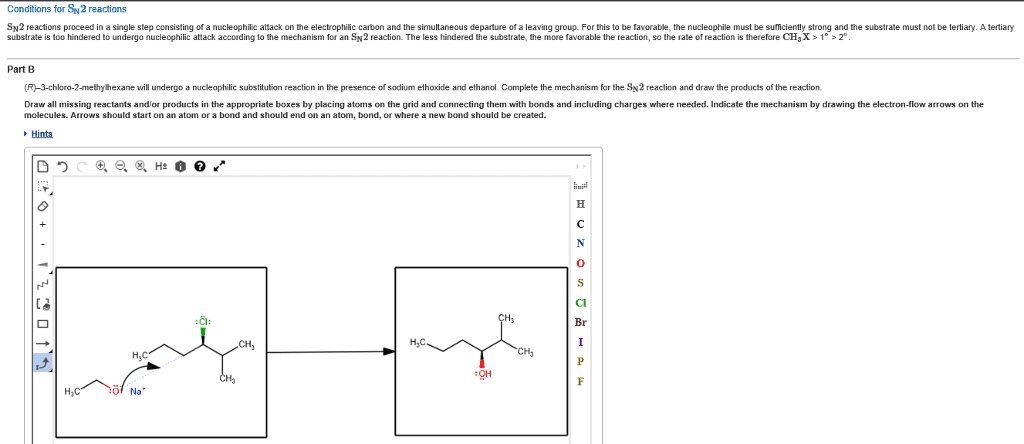
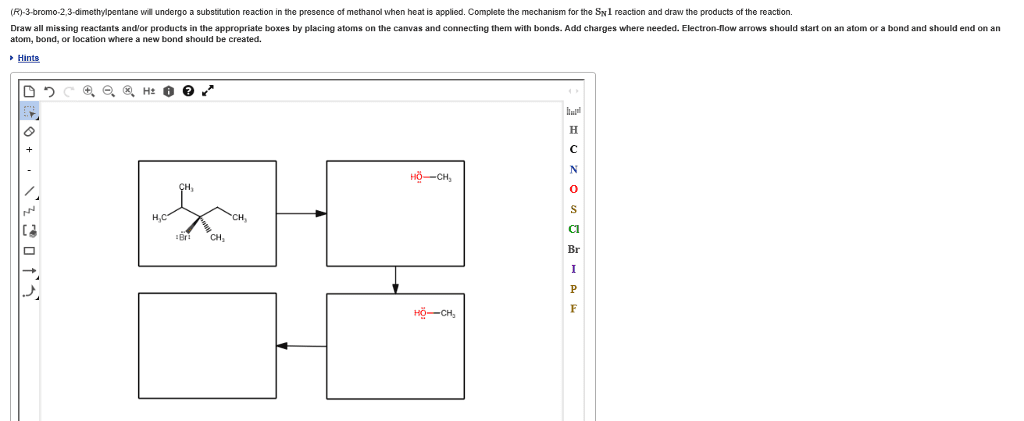
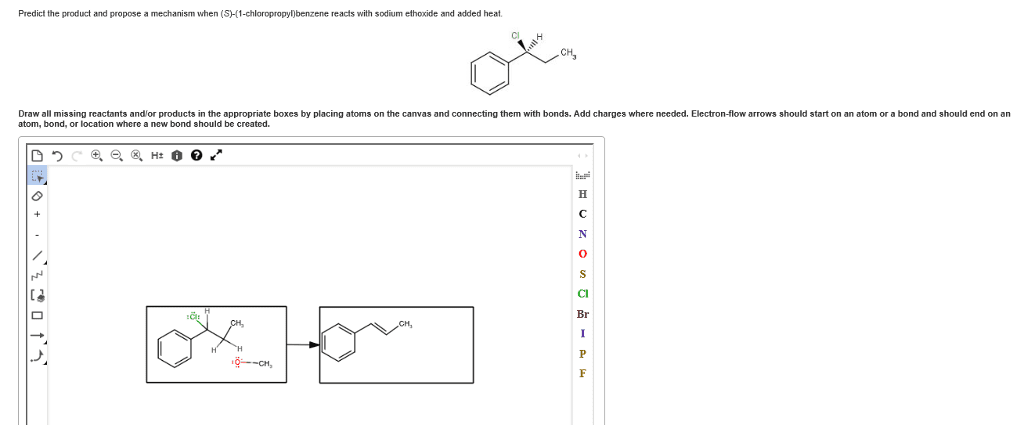
CHEM 2OA3 Chapter Notes - Chapter 7: Leaving Group, Nucleophile, Thiol
Document Summary
Substitution: exchange on group for another: occurs when a suitable electrophile + (substrate) is treated with a nucleophile . Water = weak nucleophile, h2s is a strong nucleophile (large atom, electron density is polarizable (can move around within the atom) excellent nucleophile. Electrophile (substrate) must have a leaving group: withdraws electron density via induction renders adjacent carbon atoms electrophilic, stabilize negative charge that may develop as a result of the leaving group separating from the substrate. Alpha position: carbon connected directly to the halogen. Beta position: carbon atoms connected to the alpha position: chapter focuses on reactions occurring at the alpha position. Alkyl halides: primary, secondary or tertiary based on the number of alkyl groups connected to the alpha position. Every substitution reaction exhibits at least two of the four patterns: rearrangement, proton transfer, nucleophilic attack, loss of a leaving group.