CHEM 112 Chapter Notes -Formal Charge, Lewis Structure, Covalent Bond
52 views6 pages
13 Oct 2014
School
Department
Course
Professor
Document Summary
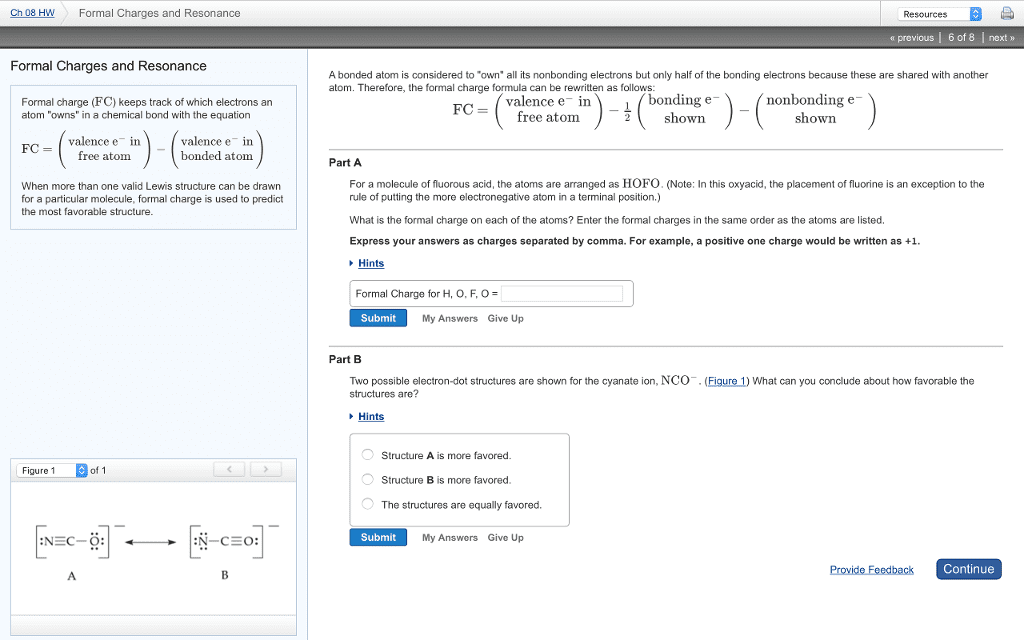
In an ideal covalent bond, electrons are exactly equally shared. Thus, we can imagine that half the electrons in each bond live on each of the two atoms participating in that bond at any given time. This leads to a very simple formula for the charge carried by an atom: we can easily determine the number of valence electrons of a neutral atom. If we can gure out whether an atom in a molecule has more or less valence electrons than the neutral atom, then we immediately know its charge. In other words, the di erence between the number of valence electrons of the neutral atom and the number of electrons it carries in a molecule is an estimate of its charge: Note that the sum of the formal charges in a molecular species is equal to the total charge, zero for a neutral molecule. Example 1. 1 the lewis diagram of carbon dioxide is.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232