CHEM 29402 Chapter Notes - Chapter 17: Chemical Equilibrium, Equilibrium Constant, Reaction Rate
50 views2 pages
13 Feb 2016
School
Department
Course
Professor
Document Summary
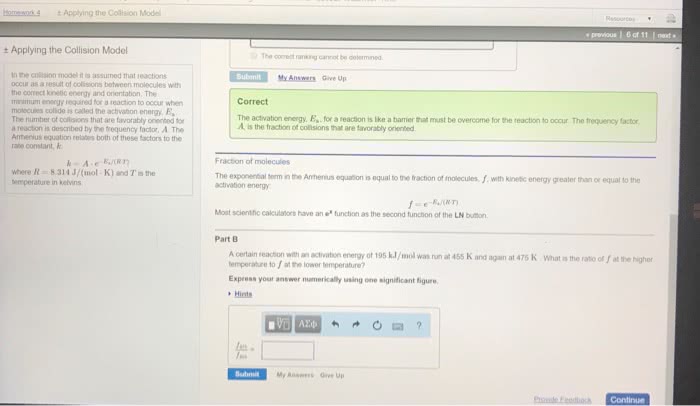
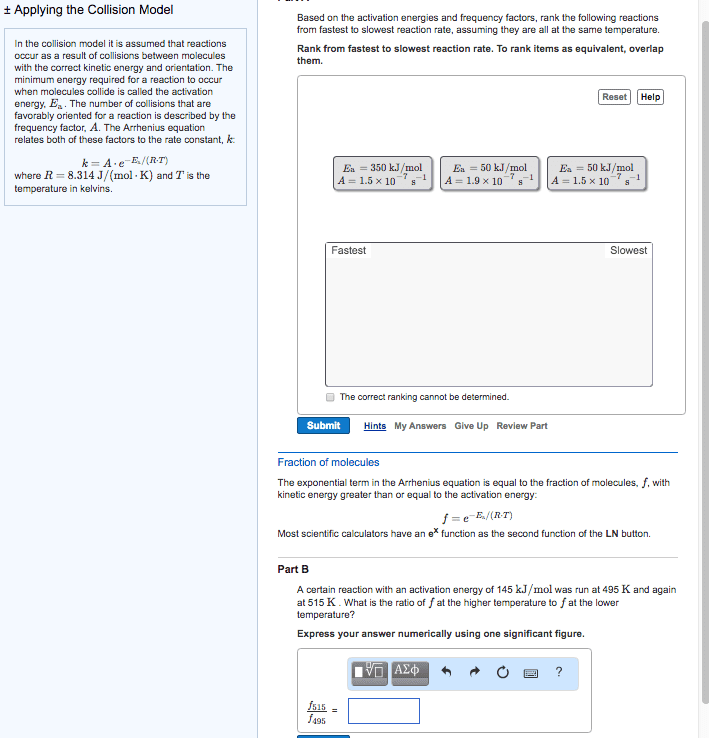
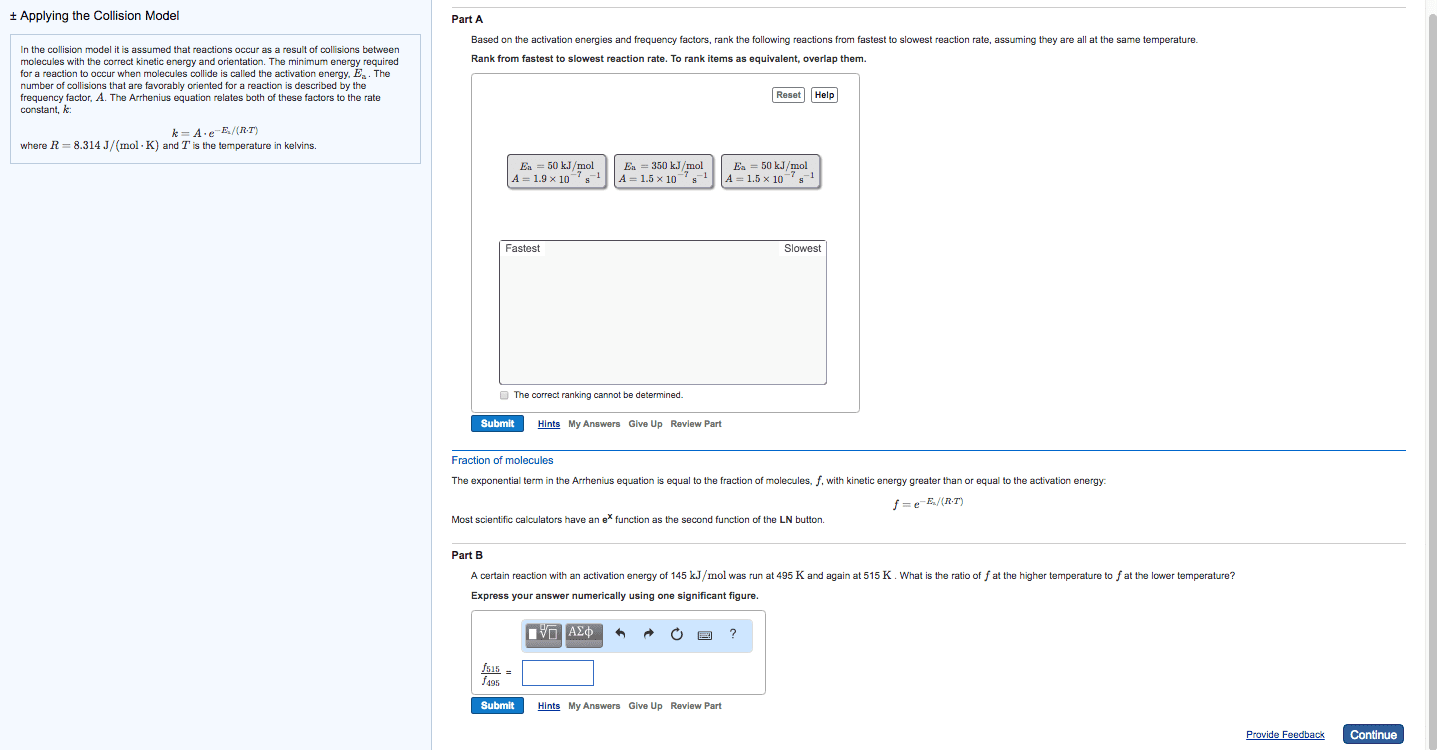
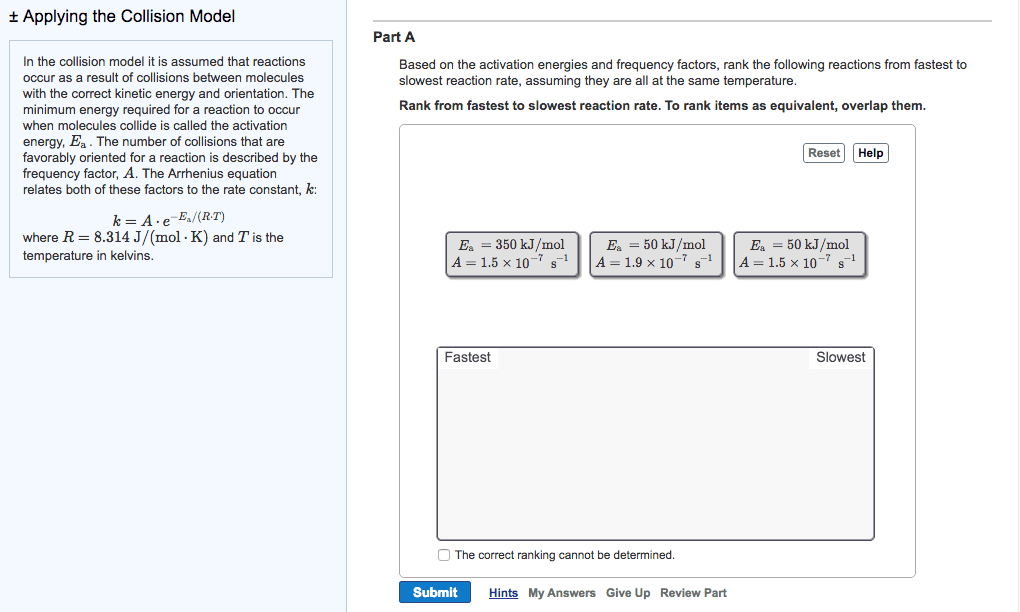
Therefore, the reaction speeds up: the minimum energy required a reaction to occur is called the activation energy (ea) If a given collision posses an energy, which is greater than the ea, then the collision can result in a reaction. If the temperature gets increased, the speeds of the molecules increase. This increases the reaction rate: catalyst: a substance that speeds up a reaction without being consumed. In the human body, enzymes act as catalysts. Enzymes allow our bodies to speed up complicated reactions. But it can be included in if it"s a gas, as the concentration of water vapour can change. Le chatelier"s principle: le chatelier"s principle states that when a change is imposed on a system as equilibrium, the position of the equilibrium shifts in a direction that tends to reduce the effect of that change. Or: when a reactant / product is added to a system at equilibrium, it shifts away from the added component.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232