CHEM102 Chapter Notes - Chapter 16.3: Rate Equation, Pressure Measurement, Reaction Rate Constant

32
CHEM102 Full Course Notes
Verified Note
32 documents
Document Summary
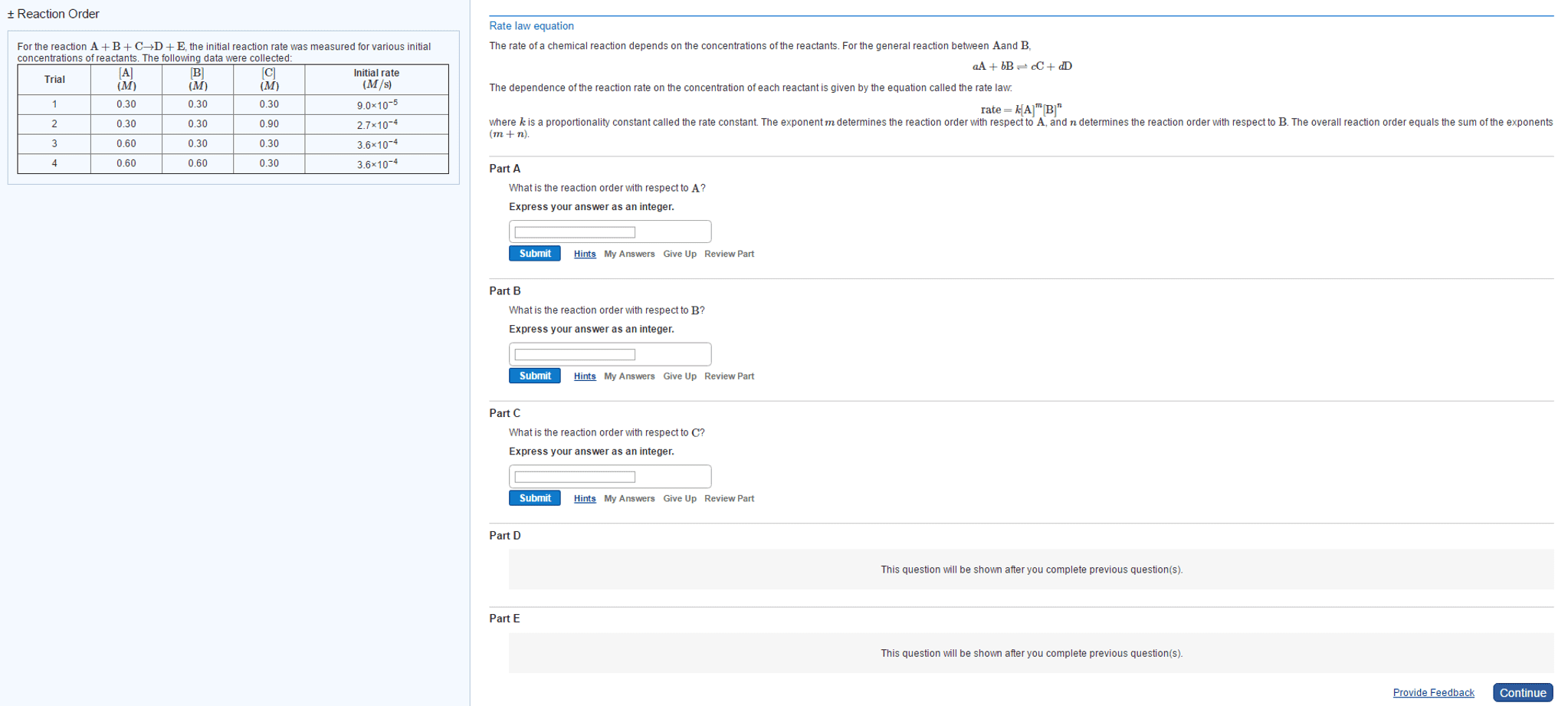
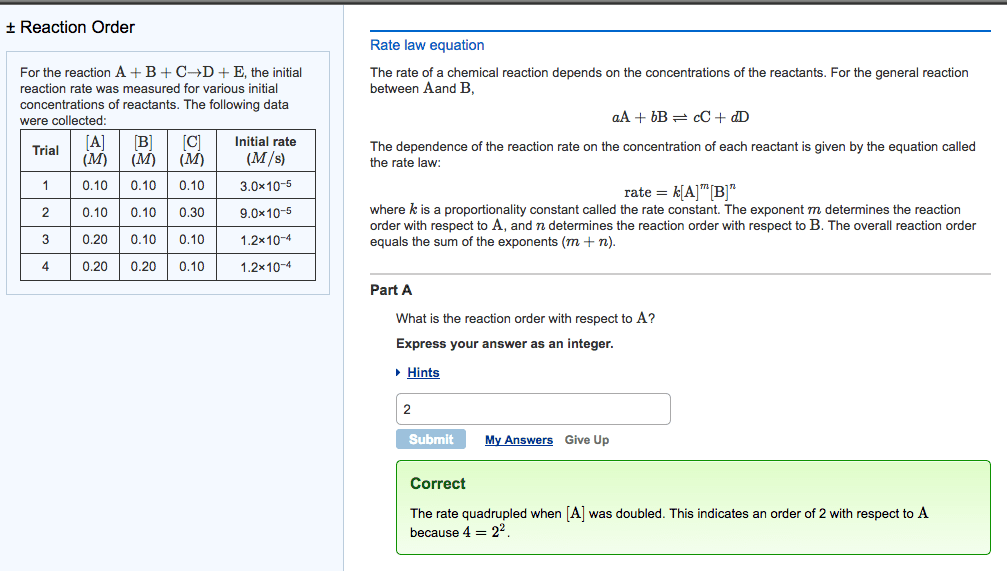
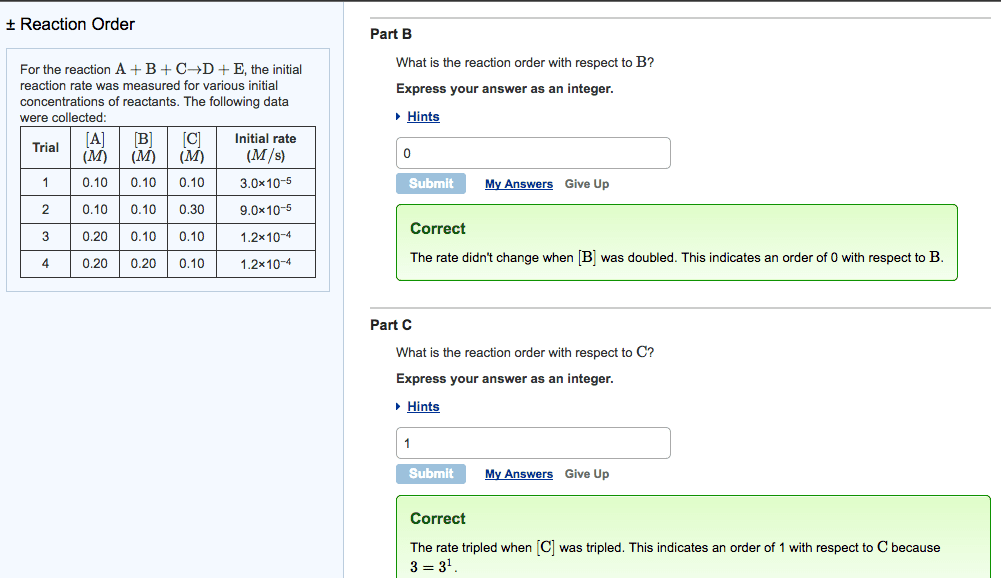
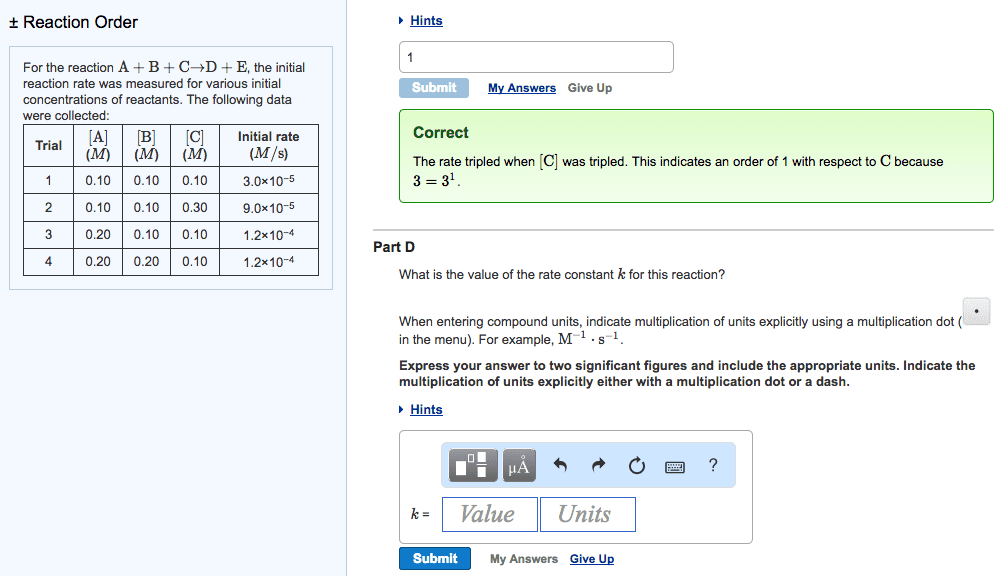
Chemistry 16. 3 the rate law and its components. Expresses the rate as a function of concentrations and temperature. K is specific for a given reaction at a given temperature and does not change as the reaction proceeds. Exponents m and n are the reaction orders define how the rate is affected by reactant concentration. Lab methods for determining initial rate: spectometric methods. Measure the concentration of a component that absorbs or emits characteristic wavelengths of light. Rate of formation is proportional to the colour intensity over time: conductometric methods. Rely on the change in electrical conductivity of reaction solution when non-ionic reactants form ionic products or vice versa: manometric methods. Manometer attached to reaction vessel of fixed volume and temperature. Manometer measures the pressure over time of a reaction that involves a change in the number of moles of gas. The rate is directly proportional to the increase in gas pressure.