CHEM105 Chapter Notes -Conjugate Acid, Weak Base, Rate Equation

15
CHEM105 Full Course Notes
Verified Note
15 documents
Document Summary
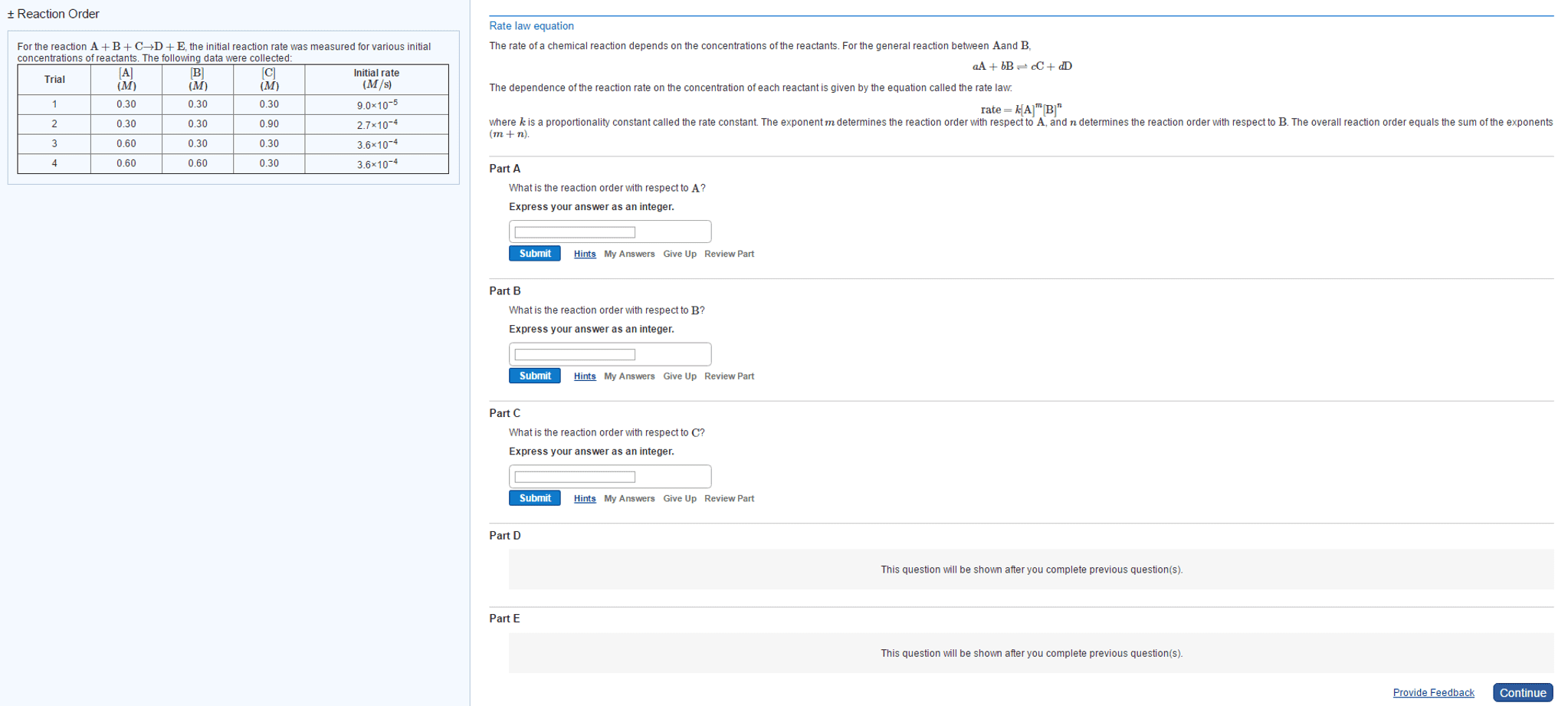
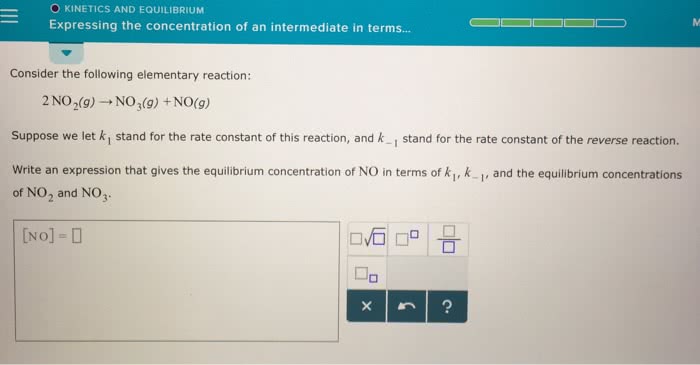
Then the reaction rate in terms of concentration can be expressed as: aa+bb cc+dd. The components of the rate law must be found by experiment, it isn"t possible to use reaction stoicheometry. This specific equilibrium constant highlights only the species whose concentrations change to any significant extent. It follows the general products over reactants rule. Like the other equilibrium constants, it is temperature dependant. Classifying the relative strengths of acids and bases: Strong bases: which are water soluble compounds containing o2- or oh-: these are either group one or group two metals which are bonded to either of those two ions. *mgo and mg(oh)2 are only slightly soluble in water, but the soluble portion dissociates completely. Weak bases: many compounds with an electron rich nitrogen atom are weak bases. A common structural feature is that the nitrogen atom has a lone pair. 18. 2 autoionization of water and the ph scale.