CHM110H5 Chapter Notes - Chapter 14: Activated Complex, Reaction Mechanism, Transition State Theory
Document Summary
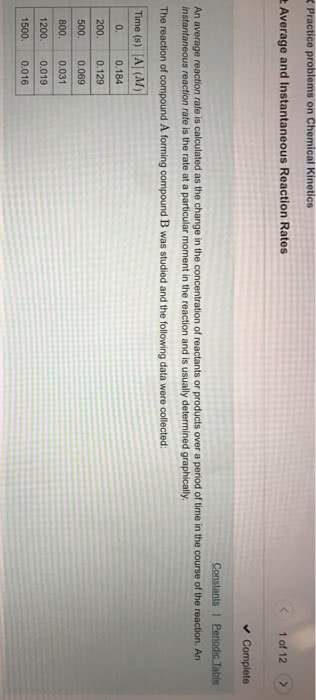
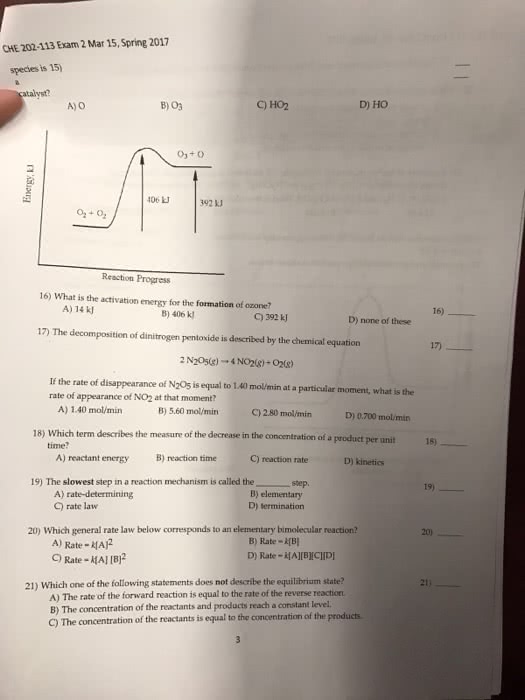
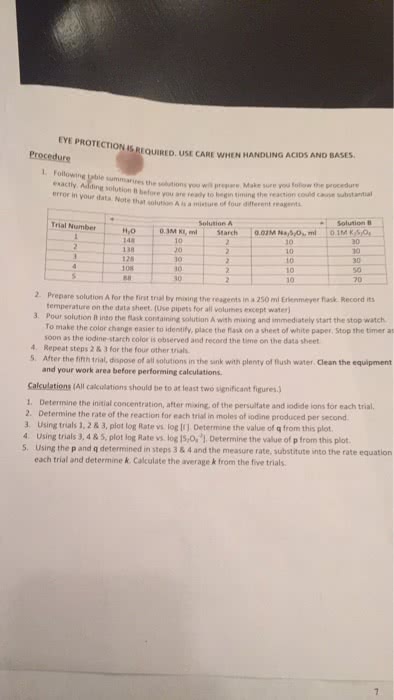
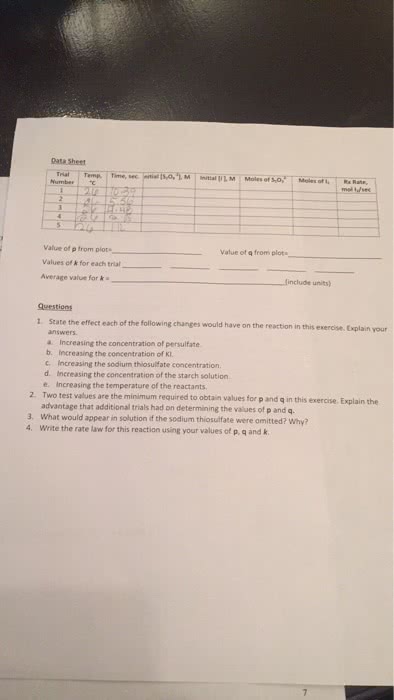
The rate of a reaction describes how fast the concentration of a reactant or product changes with time. Rate of formation = [c]/ t: rate of disappearance is a negative quantity because concentration decreases with time. Rate of disappearance = - [a]/ t: the concentration at the end of a time period is less than t was at the start of the period. To determine rate of reaction, we need to measure changes in concentration over time: reaction rate is not constant; the lower the remaining concentration of the reactant, the more slowly the reaction proceeds. Instantaneous rate of reaction- is the exact rate of a reaction at some precise point in the reaction. It is obtained from the slope of a tangent line to a concentration-time graph. Initial rate of reaction- is the rate of a reaction immediately after the reactants are brought together.