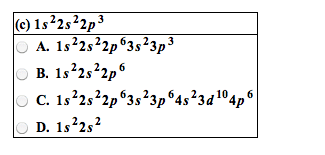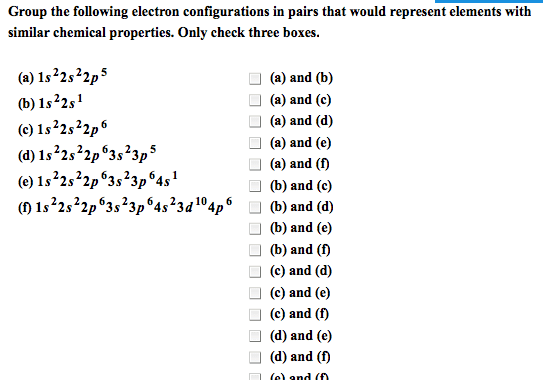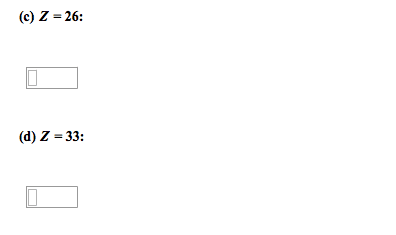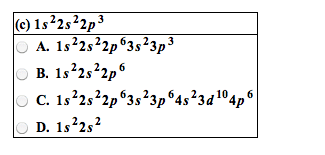
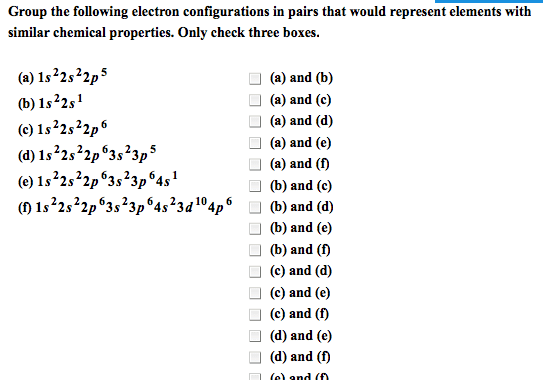

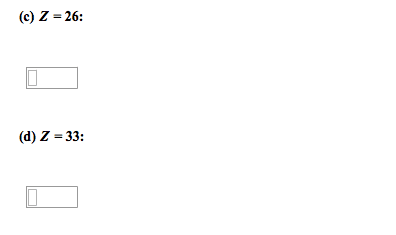






Identify the outer electron configurations for the (a) alkali metals, (b) alkaline earth metals, (c) halogens, (d) noble gases. (a) Alkali metal (b) Alkaline earth (c) Halogens (d) Noble gases metals A. ns1 A. ns1 A. ns1 A. ns1 B. ns2 B. ns2 B. ns2 B. ns2 C. ns2npl C. ns2np1 C. ns2npl C. ns2np1 D. ns2np2 D. ns2np2 D. ns2np2 D. ns2np2 E. ns2np3 E. ns2np3 E. ns2np3 E. ns2np3 F. ns2np4 F. ns2np4 F. ns2np4 F. ns2np2 G. ns2np5 G. ns2np5 G. ns2np5 G. ns2np5 H. ns2np6 H. ns2np6 H. ns2np6 H. ns2np6 For each electronic configuration given, choose the electronic configuration of the element that would match its chemical properties. Group the following electron configurations in pairs that would represent elements with similar chemical properties. Only check three boxes. Without referring to a periodic table, write the electron configuration of the elements with the following atomic numbers. Z = 9: Z = 20: Z = 26: Z = 33: A M2+ ion has four electrons in the 3d subshell and is derived from a metal in the first transition metal series. What element might M be? Elemental symbol: Write ground-state electron configurations for these ions, which play important roles in biochemical processes in our bodies. (Use a noble gas core abbreviation.) Na+ Mg2+ cl- K+ Within each group of four atoms or ions presented below, select the species that are isoelectronic with each other: Drag each item to the correct box. Arrange the following atoms in order of decreasing atomic radius: Cs K Mg A1 Na Cl P Classify the following bonds as covalent, polar covalent, or ionic. the bond in CsCl covalent polar covalent ionic the bonds in H2S covalent polar covalent ionic the NN bond in H2NNH2 covalent polar covalent ionic Draw the Lewis structure of sulfur tetrafluoride (SF4).