CHMA10H3 Chapter Notes - Chapter 18: Rate Equation, Collision Theory, Reaction Rate

36
CHMA10H3 Full Course Notes
Verified Note
36 documents
Document Summary
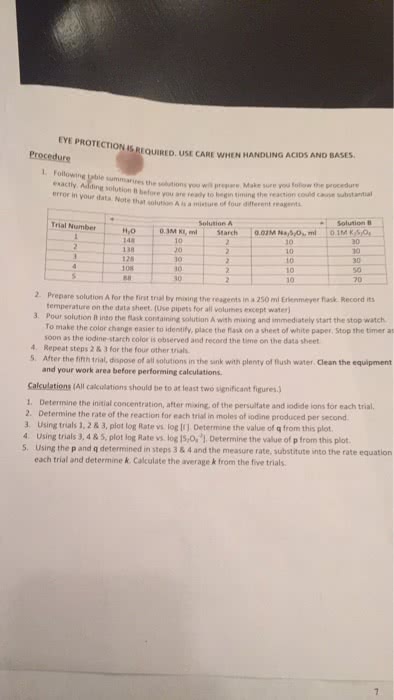
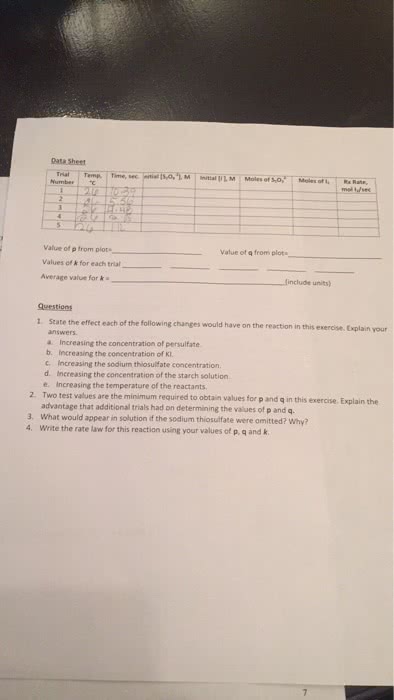
Chemical kinetics is the study of the rates of chemical reactions. The rate of a chemical reaction is measured as the change in concentration of a reactant or product (which must be specified) per unit of time. The average rate over a given time duration and the instantaneous rate at any moment must be distinguished. The concept of reaction rate - decomposition of n2o5 . Conditions that affect the rate of a reaction. The rate of a given reaction can be increased by. An increase in surface area, if the reactant is solid. For a reaction of the general form: aa + bb xx. The dependence of reaction rate on the concentrations of reactants is expressed by a rate equation of the form. The values of the exponents m and n are not necessarily the same as the stoichiometric coefficients, and can only be determined experimentally.