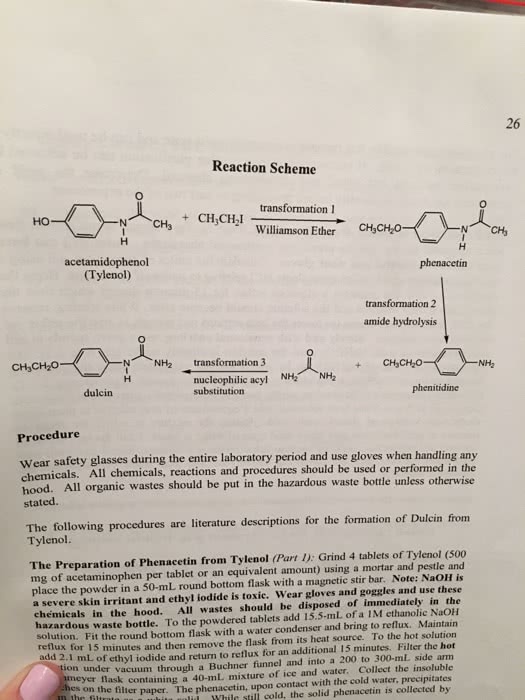
CHMB16H3 Chapter 3: CHMB16 Chapter 3
Document Summary
Treat spills on your skin first by flooding with water. The lab notebook must state what was done, what was observed and be understandable to someone else: use complete sentences to prevent incomplete descriptions. Paste hard copies of important data into your notebook. Each page in your notebook should be signed and dated. Electronic balance: uses electromagnetic force compensation to balance the load on the pan. It should be in its arrested position when you load or unload the pan and in the half-arrested position when you are dialing weights. For best precision, measure the mass of reagent, instead of volume, delivered from a burette or syringe. Ignition: used to convert a precipitate into a known, constant composition. it because it leaves little residue when burned. Slurry: suspension of solid in liquid (pour the slurry of precipitate down a glass rod to prevent splattering): rubber policeman: a flattened piece of rubber at the end of a glass rod.