CHMB20H3 Chapter 2: CHMB20 Chapter 2
Document Summary
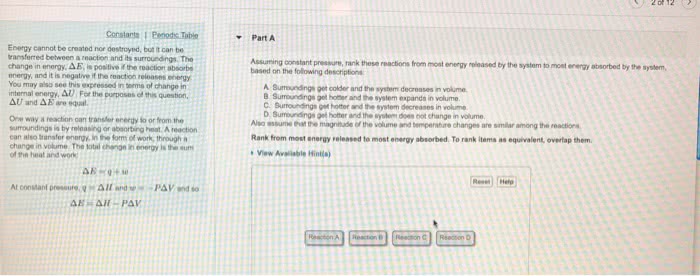
Thermodynamics: the study of the transformations of energy. System: the part of the world in which we have a special interest. Isolated system: a closed system that has neither mechanical nor thermal contact with its surroundings. system and its surroundings: work: done to achieve motion against an opposing force; the transfer of energy that makes use of organized motion. Endothermic and diathermic container results in energy flowing into the system as heat to restore the temperature to that of the surroundings. Exothermic and diathermic container results in a release of energy as heat into the surroundings. Endothermic and adiabatic container results in a lowering of temperature of the system. Exothermic and adiabatic results in a rise of temperature of the system. Thermal motion: disorderly motion of molecules in the surroundings (movement of heat). For monatomic gas (translation only), um(t) = um(0) + (3/2)rt, where. Um(-) is the molar internal energy at t = 0.