The images represent three samples of nitrogen dioxide, which is the pollutant responsible for the brown color of the atmospheric pollution. At high temperatures, nitrogen dioxide decomposes to nitrogen monoxide and oxygen gas.
Use the particulate model below to answer the following questions.
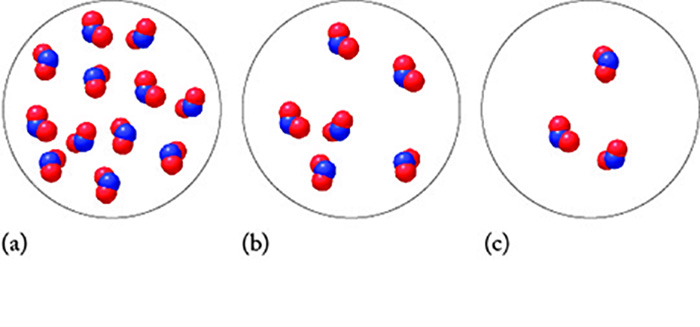
Part 1 Which statement is true regarding the rate constant for models (a), (b), and (c)? Assume equal temperatures and volumes for all three models.
Choose one: A. Model (a) has the largest rate constant. B. Model (b) has the largest rate constant. C. Model (c) has the largest rate constant. D. The rate constant is equal in (a), (b), and (c).
Part 2 Which model (a), (b), or (c) has the largest instantaneous rate of reaction? The reaction is not zero order.
Choose one: A. Model (a) B. Model (b) C. Model (c) D. The instantaneous rate is equal in models (a), (b), and (c). E. Insufficient information is provided to predict the relationship.
Part 3 A plot of 1/NO2 versus time is linear. Using this information, identify the factor by which the rate will increase in model (c) if the number of molecules is increased by a factor of 20.


