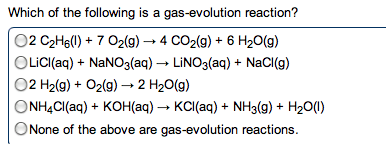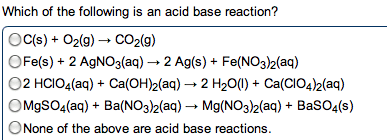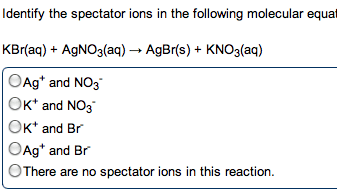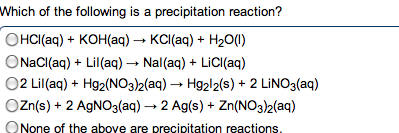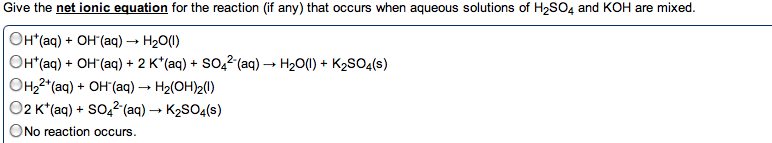CHM135H1 Chapter Notes - Chapter 16: Chemical Equation, Weak Base, Conjugate Acid
100 views2 pages

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
Chm135 chapter 16 study notes (aq) (aq) + oh- Example: hcl(aq) + naoh(aq) h2o(l) + nacl(aq) Hcl, naoh, nacl completely dissociated, net ionic equation h3o+ If we mix equal numbers of moles of hcl and naoh, concentrations of h3o+ and oh- remaining in nacl after neutralization same as pure water, [h3o+] = [oh-] = 1. 0 x 10-7m. Equilibrium constant (kn): kn = 1/kw (kn = 1. 0 x 1014) Value for strong acid-strong base reaction very large, proceeds 100% to completion. After neutralization of equal molar amounts acid and base, solution contains salt derived. Because neither cation nor anion of salt has acidic or basic properties, ph is 7. Because weak acid largely undissociated, net ionic equation for reaction of weak acid with strong base involves proton transfer from ha to strong base: Ha(aq) + oh- (aq) h2o(l) + a- (aq) (aq) (aq) + b(aq) h2o(l) + bh+