Chemistry 1027A/B Chapter Notes - Chapter 1.2: Reaction Quotient, Solubility Equilibrium, Supersaturation
Document Summary
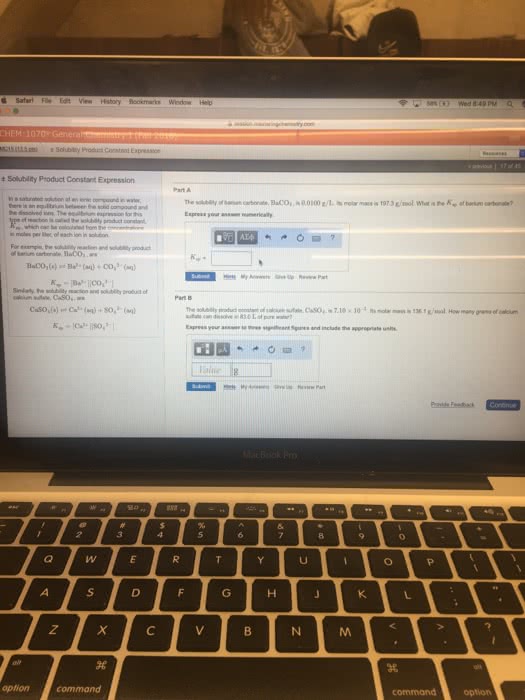
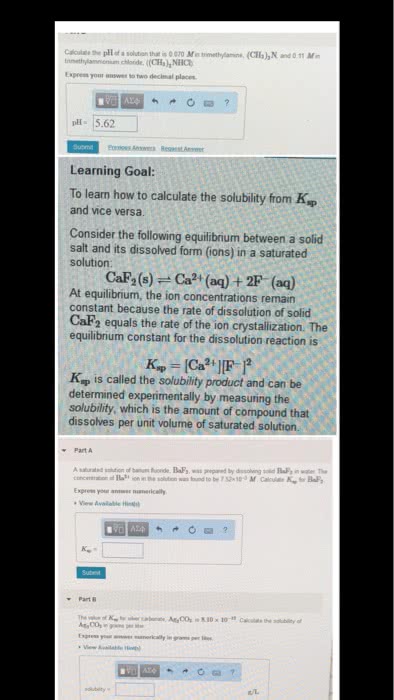
Solubility is the quantity of a compound that will dissolve in a certain amount of solution. The solubility product or solubility product constant (ksp) is an equilibrium constant. Saturated means that the maximum amount of compound has dissolved. Unsaturated means that there is no compound remaining, but it is possible for more to dissolve. Supersaturated means that more than the maximum amount of compound has dissolved, thus a. Axby (s) xay+ (aq) + ybx- (aq) The reaction quotient, or ion product (q) is the same as the solubility product, except the concentrations used do not have to be at equilibrium. When q < ksp no solid will be present and all ions will remain in solution: the reaction will proceed to the right. When q = ksp the system is at equilibrium (saturated) When q > ksp a solid will be formed from the ions in solution, which is a precipitate reaction (supersaturated: the reaction will proceed to the left.