Chemistry 1302A/B Chapter Notes - Chapter 3.4: Conjugate Acid, Acid Strength, Equivalence Point

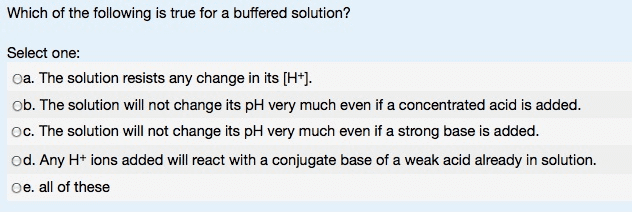
Buffer Solution:
Must contain weak acid that will react with any
OH#
ions (from a strong
base) that may be added to the solution
1.
Must also contain a weak base that will react with any
H$O%
ions (from a
strong acid) that may be added to a sloution
2.
The acid and the base in the buffer solution must not react with each
other
The only species that can possibly qualify are a weak species and its
own conjugate partner
§
The weak species and its conjugate partner must both be present in
significant amounts
§
Work best when there is a about a 1:1 mixture of the weak species
and its conjugate, but the solution will still perform its buffering
function if the 2 concentrations are within a factor of 10
§
3.
Capable of resisting changes in pH when small amounts of strong species
are added to it
○
•
If a solution contains both a weak acid and its conjugate base, they must be in
equilibrium
•
A buffer solution consists of a weak species with its conjugate partner behaving
as a common ion
•
Preparing a Buffer Solution
If a buffer solution contains a weak acid and its conjugate base, the pH of that
buffer will generally fall in the acidic range
•
If a buffer solution contains a weak base and its conjugate acid, the pH of that
buffer will generally fall in the basic range
•
Acid Buffer
Acid Buffer: buffer solution composed of a weak acid and its conjugate base
•
Easiest way to prepare is to start with a solution of weak acid and add a salt that
contains the conjugate base
•
Also can start with either the weak acid or its conjugate base and use a strong
species to create the other
Correct amount of strong species will convert roughly half of the weak
species to its conjugate species
○
•
Base Buffer
Base Buffer: buffer solution composed of a weak base and its conjugate acid•
Easiest way to prepare is to start with a solution of weak base and add its
conjugate acid
•
Also can start with either the weak base or its conjugate acid and use a strong
species to create the other
Correct amount of strong species will convert roughly half of the weak
species to its conjugate species
○
•
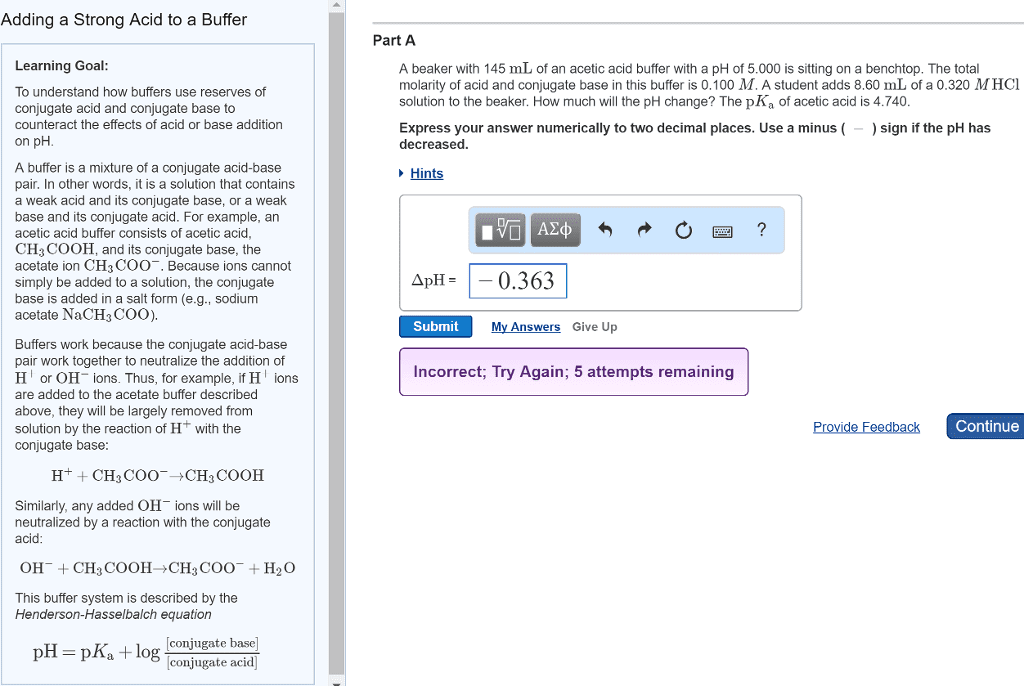
pH of a Buffer Solution
pH of a buffer depends only on the ratio of the concentrations of the two
components, so it is unaffected by dilution
•
Buffer solution has a strong ability to resist changes in pH•
The value of 𝑥is always neglible when compared to the concetration of the
parent or conjugate species
•
Can use mole amounts of the parent and conjugate species instead of
concentration for equilibrium because both have the same volume since they
are in the same solution
H$O%& OH#must always be in concentration
○
•
Acid-Base Titration
Acid-Base titration: method used to determine parameters such as the
concentration of a base or acid in a solution
•
2 possibilities in a titration process:
Acid is added to neutralize a base
○
Base is added to neutralize an acid
○
•
Trnt of a titration is reached when stoichiometrically equivalent amounts of acid
and base have been combined
•
The products of the ionization reaction of the salt determines whether the final
solution is acidic or basic
•
pH at Equivalence Point
For any titration involving a weak species and a strong species, an equilibrium
will be established between the excess weak species and the just-formed
conjugate species
Buffer solution
○
At the equivalence point, the only species present is the conjugate, which
will then react with water
Responsible for the pH of the solution at equivalence point
§
○
•
Acid-Base Indicators
Used to detect the equivalence point in a titration•
An indicator is a weak organic acid (HIn) that has a different colour than its
conjugate base
The indicator is a different colour depending if its in an acidic solution or
basic solution
○
The colour change takes place around when the pH value where
[HIn]=[In#] (endpoint)
pK+,- =pH when [HIn]=[In#]
§
○
•
Titration Equivalence Point pH Desired indicator (pK/)Typical Indicator
Strong acid-
strong base
Neutral 7 Bromothymol blue
Strong acid-
weak base
Acidic 4-6 Methyl red
Weak acid-
strong base
Basic 8-10 phenolphthalein
Titration Curves
Shows the pH resulting from all the reactions occurring during a titration•
Strong Acid-Strong Base Titration
•
Weak Acid-Strong Base Titration
•
Weak Base-Strong Acid Titration
•
3.4 Buffers and Titrations
Thursday, December 21, 2017
1:22 AM

Buffer Solution:
Must contain weak acid that will react with any OH#ions (from a strong
base) that may be added to the solution
1.
Must also contain a weak base that will react with any H$O%ions (from a
strong acid) that may be added to a sloution
2.
The acid and the base in the buffer solution must not react with each
other
The only species that can possibly qualify are a weak species and its
own conjugate partner
§
The weak species and its conjugate partner must both be present in
significant amounts
§
Work best when there is a about a 1:1 mixture of the weak species
and its conjugate, but the solution will still perform its buffering
function if the 2 concentrations are within a factor of 10
§
3.
Capable of resisting changes in pH when small amounts of strong species
are added to it
○
•
If a solution contains both a weak acid and its conjugate base, they must be in
equilibrium
•
A buffer solution consists of a weak species with its conjugate partner behaving
as a common ion
•
Preparing a Buffer Solution
If a buffer solution contains a weak acid and its conjugate base, the pH of that
buffer will generally fall in the acidic range
•
If a buffer solution contains a weak base and its conjugate acid, the pH of that
buffer will generally fall in the basic range
•
Acid Buffer
Acid Buffer: buffer solution composed of a weak acid and its conjugate base•
Easiest way to prepare is to start with a solution of weak acid and add a salt that
contains the conjugate base
•
Also can start with either the weak acid or its conjugate base and use a strong
species to create the other
Correct amount of strong species will convert roughly half of the weak
species to its conjugate species
○
•
Base Buffer
Base Buffer: buffer solution composed of a weak base and its conjugate acid•
Easiest way to prepare is to start with a solution of weak base and add its
conjugate acid
•
Also can start with either the weak base or its conjugate acid and use a strong
species to create the other
Correct amount of strong species will convert roughly half of the weak
species to its conjugate species
○
•
pH of a Buffer Solution
pH of a buffer depends only on the ratio of the concentrations of the two
components, so it is unaffected by dilution
•
Buffer solution has a strong ability to resist changes in pH•
The value of 𝑥is always neglible when compared to the concetration of the
parent or conjugate species
•
Can use mole amounts of the parent and conjugate species instead of
concentration for equilibrium because both have the same volume since they
are in the same solution
H$O%& OH#must always be in concentration
○
•
Acid-Base Titration
Acid-Base titration: method used to determine parameters such as the
concentration of a base or acid in a solution
•
2 possibilities in a titration process:
Acid is added to neutralize a base
○
Base is added to neutralize an acid
○
•
Trnt of a titration is reached when stoichiometrically equivalent amounts of acid
and base have been combined
•
The products of the ionization reaction of the salt determines whether the final
solution is acidic or basic
•
pH at Equivalence Point
For any titration involving a weak species and a strong species, an equilibrium
will be established between the excess weak species and the just-formed
conjugate species
Buffer solution
○
At the equivalence point, the only species present is the conjugate, which
will then react with water
Responsible for the pH of the solution at equivalence point
§
○
•
Acid-Base Indicators
Used to detect the equivalence point in a titration•
An indicator is a weak organic acid (HIn) that has a different colour than its
conjugate base
The indicator is a different colour depending if its in an acidic solution or
basic solution
○
The colour change takes place around when the pH value where
[HIn]=[In#] (endpoint)
pK+,- =pH when [HIn]=[In#]
§
○
•
Titration Equivalence Point pH Desired indicator (pK/)Typical Indicator
Strong acid-
strong base
Neutral 7 Bromothymol blue
Strong acid-
weak base
Acidic 4-6 Methyl red
Weak acid-
strong base
Basic 8-10 phenolphthalein
Titration Curves
Shows the pH resulting from all the reactions occurring during a titration•
Strong Acid-Strong Base Titration
•
Weak Acid-Strong Base Titration
•
Weak Base-Strong Acid Titration
•
3.4 Buffers and Titrations
Thursday, December 21, 2017
1:22 AM

Buffer Solution:
Must contain weak acid that will react with any OH#ions (from a strong
base) that may be added to the solution
1.
Must also contain a weak base that will react with any H$O%ions (from a
strong acid) that may be added to a sloution
2.
The acid and the base in the buffer solution must not react with each
other
The only species that can possibly qualify are a weak species and its
own conjugate partner
§
The weak species and its conjugate partner must both be present in
significant amounts
§
Work best when there is a about a 1:1 mixture of the weak species
and its conjugate, but the solution will still perform its buffering
function if the 2 concentrations are within a factor of 10
§
3.
Capable of resisting changes in pH when small amounts of strong species
are added to it
○
•
If a solution contains both a weak acid and its conjugate base, they must be in
equilibrium
•
A buffer solution consists of a weak species with its conjugate partner behaving
as a common ion
•
Preparing a Buffer Solution
If a buffer solution contains a weak acid and its conjugate base, the pH of that
buffer will generally fall in the acidic range
•
If a buffer solution contains a weak base and its conjugate acid, the pH of that
buffer will generally fall in the basic range
•
Acid Buffer
Acid Buffer: buffer solution composed of a weak acid and its conjugate base•
Easiest way to prepare is to start with a solution of weak acid and add a salt that
contains the conjugate base
•
Also can start with either the weak acid or its conjugate base and use a strong
species to create the other
Correct amount of strong species will convert roughly half of the weak
species to its conjugate species
○
•
Base Buffer
Base Buffer: buffer solution composed of a weak base and its conjugate acid
•
Easiest way to prepare is to start with a solution of weak base and add its
conjugate acid
•
Also can start with either the weak base or its conjugate acid and use a strong
species to create the other
Correct amount of strong species will convert roughly half of the weak
species to its conjugate species
○
•
pH of a Buffer Solution
pH of a buffer depends only on the ratio of the concentrations of the two
components, so it is unaffected by dilution
•
Buffer solution has a strong ability to resist changes in pH
•
The value of
𝑥
is always neglible when compared to the concetration of the
parent or conjugate species
•
Can use mole amounts of the parent and conjugate species instead of
concentration for equilibrium because both have the same volume since they
are in the same solution
H$O%
&
OH#
must always be in concentration
○
•
Acid-Base Titration
Acid-Base titration: method used to determine parameters such as the
concentration of a base or acid in a solution
•
2 possibilities in a titration process:
Acid is added to neutralize a base
○
Base is added to neutralize an acid
○
•
Trnt of a titration is reached when stoichiometrically equivalent amounts of acid
and base have been combined
•
The products of the ionization reaction of the salt determines whether the final
solution is acidic or basic
•
pH at Equivalence Point
For any titration involving a weak species and a strong species, an equilibrium
will be established between the excess weak species and the just-formed
conjugate species
Buffer solution
○
At the equivalence point, the only species present is the conjugate, which
will then react with water
Responsible for the pH of the solution at equivalence point
§
○
•
Acid-Base Indicators
Used to detect the equivalence point in a titration•
An indicator is a weak organic acid (HIn) that has a different colour than its
conjugate base
The indicator is a different colour depending if its in an acidic solution or
basic solution
○
The colour change takes place around when the pH value where
[HIn]=[In#] (endpoint)
pK+,- =pH when [HIn]=[In#]
§
○
•
Titration Equivalence Point pH Desired indicator (pK/)Typical Indicator
Strong acid-
strong base
Neutral 7 Bromothymol blue
Strong acid-
weak base
Acidic 4-6 Methyl red
Weak acid-
strong base
Basic 8-10 phenolphthalein
Titration Curves
Shows the pH resulting from all the reactions occurring during a titration•
Strong Acid-Strong Base Titration
•
Weak Acid-Strong Base Titration
•
Weak Base-Strong Acid Titration
•
3.4 Buffers and Titrations
Thursday, December 21, 2017 1:22 AM
Document Summary
Must contain weak acid that will react with any oh# ions (from a strong base) that may be added to the solution. Must also contain a weak base that will react with any h% ions (from a strong acid) that may be added to a sloution. The acid and the base in the buffer solution must not react with each other. The only species that can possibly qualify are a weak species and its own conjugate partner. The weak species and its conjugate partner must both be present in significant amounts. Work best when there is a about a 1:1 mixture of the weak species and its conjugate, but the solution will still perform its buffering function if the 2 concentrations are within a factor of 10. Capable of resisting changes in ph when small amounts of strong species are added to it. If a solution contains both a weak acid and its conjugate base, they must be in equilibrium.