CHEM 1A Chapter Notes - Chapter 3.3: Structural Formula, Covalent Bond, Chemical Formula
Document Summary
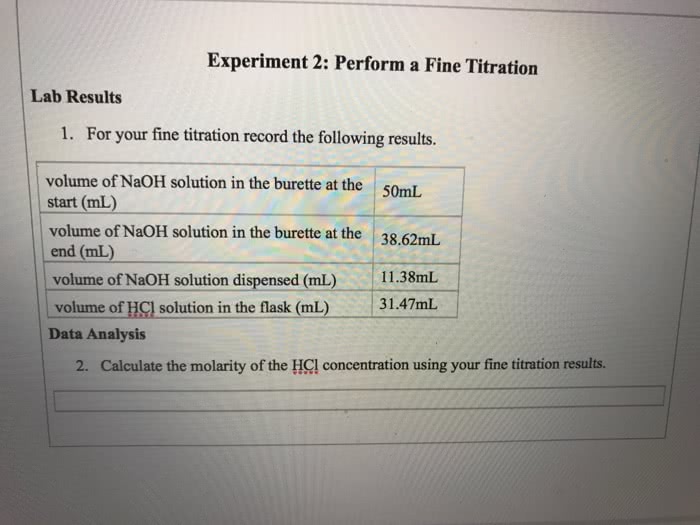
Molecular formula actual number of atoms of each element in a molecule of a empirical formula relative number of atoms in each element in a compound symbol for each element, subscript indicating relative number of atoms of element compound whole number multiple of empirical formula structural formula use lines to represent covalent bonds, show how molecules are connected or bonded to each other: can depict bond angles, double bonds ball and stick models represent atoms as balls and chemical bonds as sticks (cid:224) represent shape of molecule, use color to represent different elements space filling molecular models atoms fill space between each other represent estimate of how molecule would actually look.