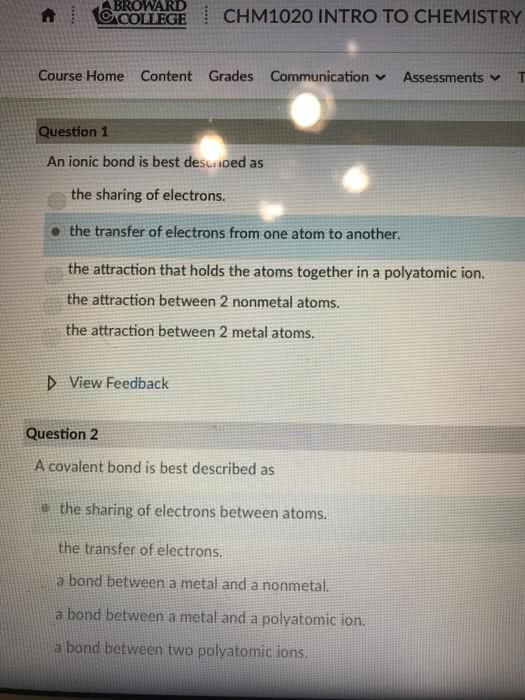
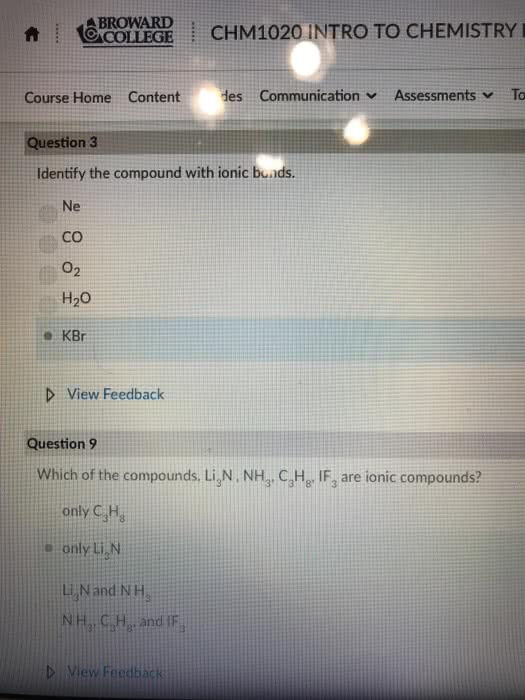
Exam 2, CHEM 141 Spring 2018, Jackson State University Please write clearly. No credit will be given for illegible or ambiguous work. Please write your name on each page Section A. Multiple Choice Questions. Choose the best answer. Each question in this section is worth 4 points. Make sure you answer all questions 1. An ionic bond is best described as a) the attraction between 2 metal atoms b) the sharing of electrons by 2 atoms e) the electrostatic attraction between oppositely charged ions. d) the attraction between 2 nonmetal atoms. e) the attraction that holds the atoms together in a polyatomic ion Identify the compound with ionic bonds. a) Srl 2. b) NO c) He d) CBrs e) PF, 3. Which pair of elements would be most likely to form an ionic compound? a. P and Br b. Cu and K c. C and O d. Al and Rb 4. Which one of the following compounds is molecular e. a. Fe O Which of the following is a molecular element? a) sodium 5. b) iron c) carbon d) copper e)hydrogen 6. 0.274 moles of a substance has a mass of 62.5 g. What is the molar mass of the a) 0.00438 g mol b) 17.1 g mol'e) 228 g mol d) 17.1 amu e) 228 amu 7. What is the charge on the Fe ions in Fe(PO)2 substance? Write the formula for the compound formed between barium and nitrogen. a) BajN2 8. b) BazN c BaN 9. Give the formula for magnesium nitrite. d) Mg(NO e) Mg N2 10. The solid compound, Sny(PO4)4, contains a) Sn,t, P*, and O ions. b) Sn" and PO ions c) Sn" and POions. d) Sny(PO4h molecules. 11. What is the empirical formula for CroHs? a) CsH