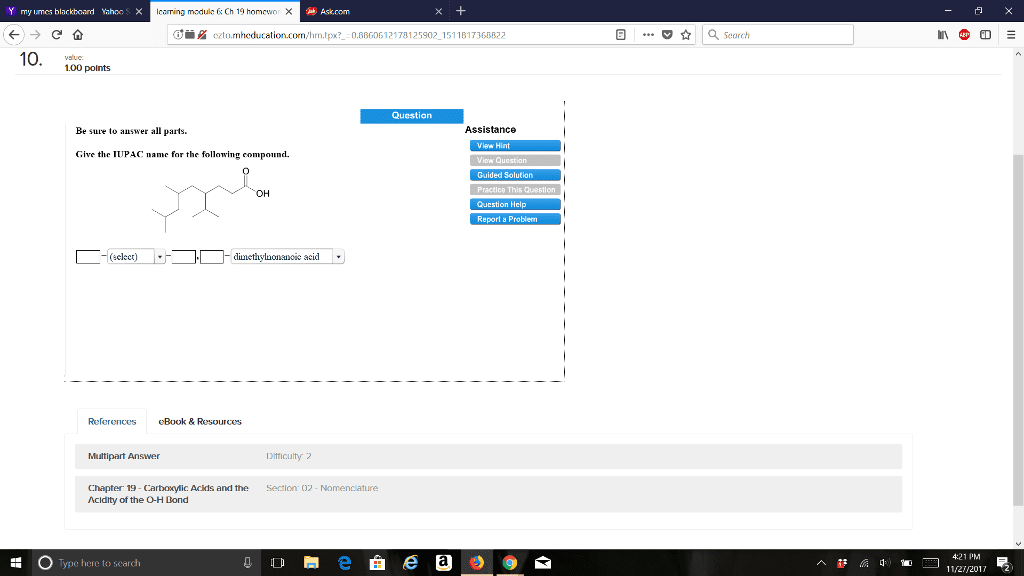
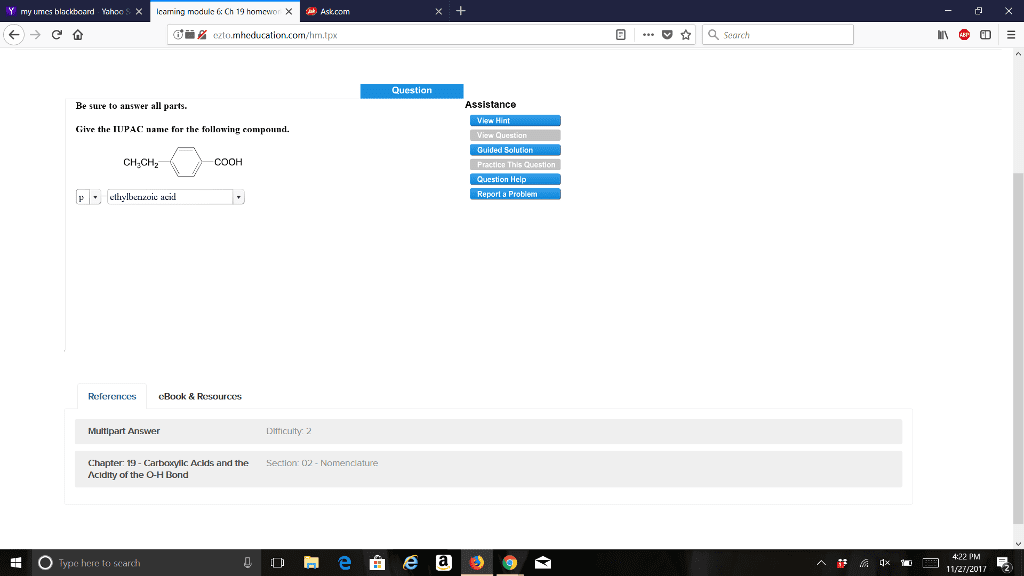
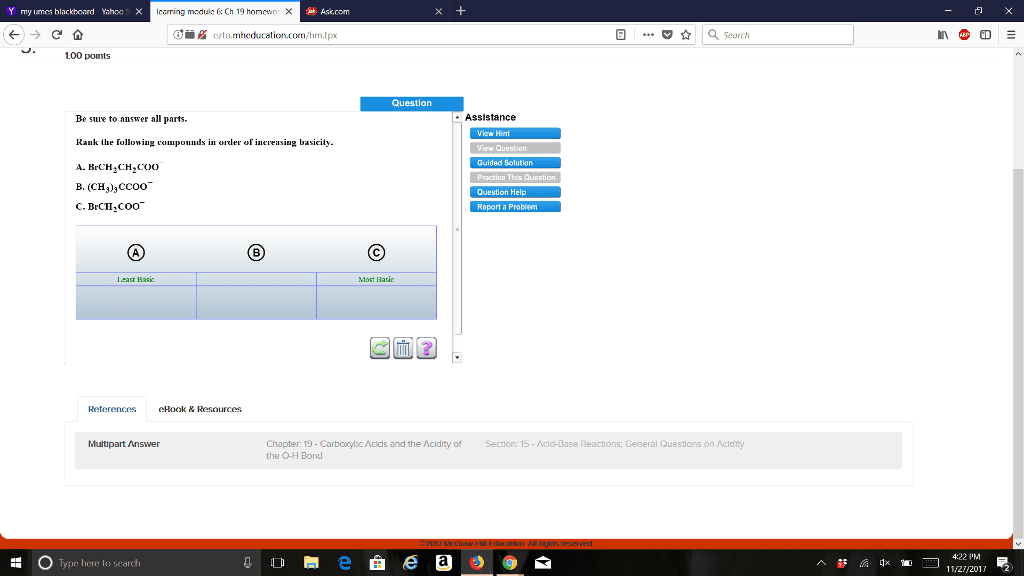
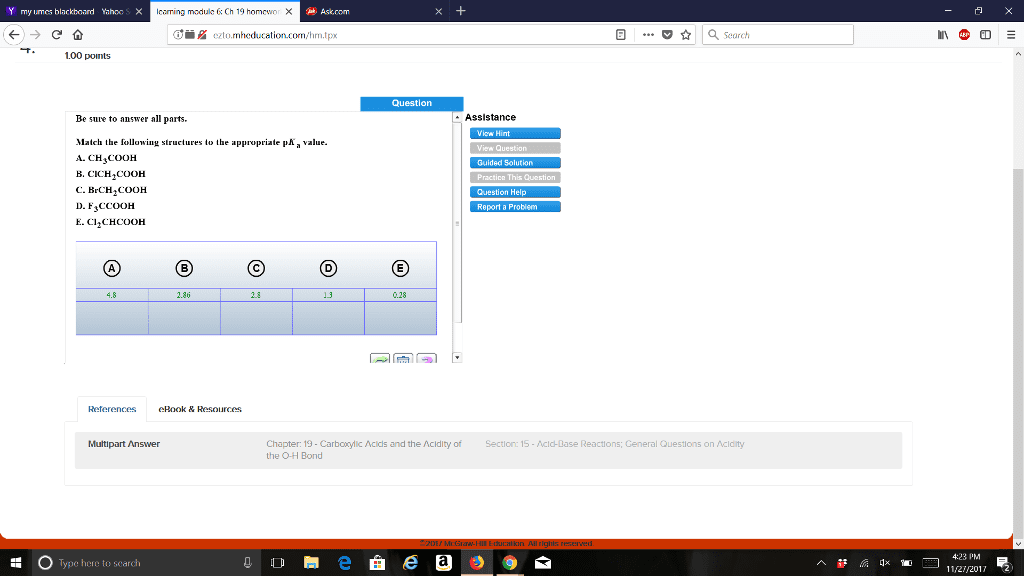
CHEM 273 Chapter Notes - Chapter 19: Sodium Chloride, Alanine, Acid Dissociation Constant
Document Summary
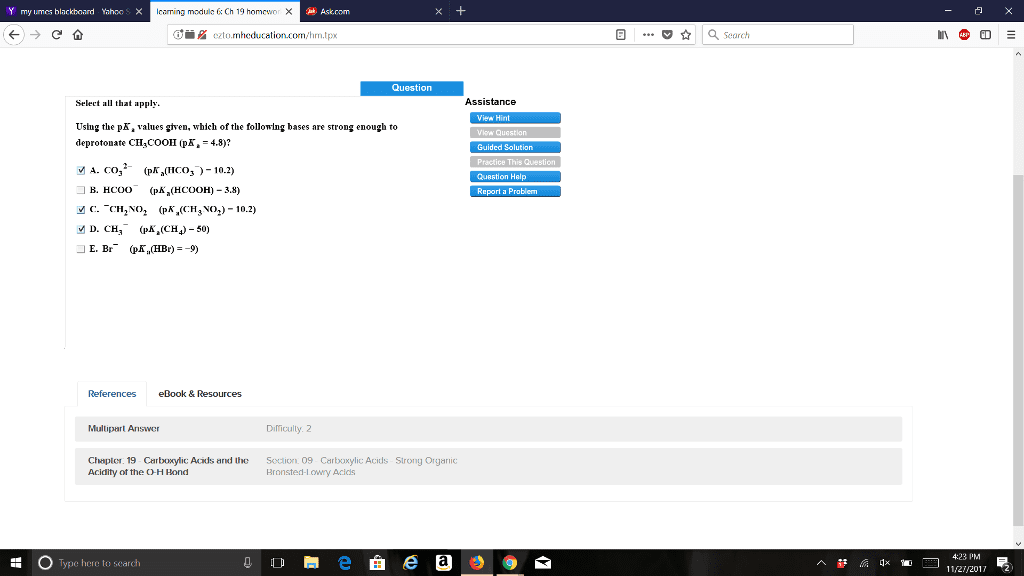
A c=o absorption at 1710 cm-1 and a c-h absorption at 3000 cm-1. A c=o absorption at 1710 cm-1 and an o-h absorption at 2500-3500 cm-1. A c=o absorption at 1600 cm-1 and an o-h absorption at 2500-3000 cm-1. A c-o absorption at 1500 cm-1 and an o-h absorption at 2500-3500 cm-1. Rank the labeled protons (ha-hd) in order of increasing acidity, starting with the least acidic. Rank the following compounds in order of decreasing acidity, putting the most acidic rst. Rank the following compounds in order of increasing acidity, putting the least acidic rst. The benzoic acid would dissolve in the water layer and the nacl would dissolve in the organic layer. The benzoic acid would dissolve in the organic layer and the nacl would dissolve in the water layer. Both benzoic acid and nacl would dissolve in the organic layer. Both benzoic acid and nacl would dissolve in the water layer.