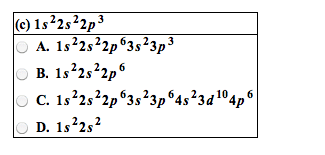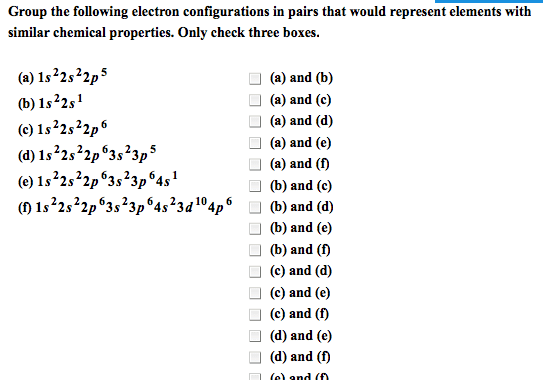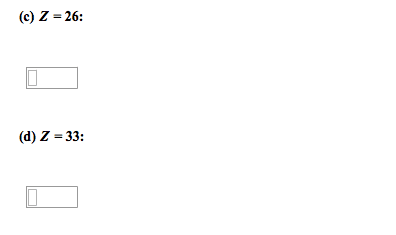CHEM 1211 Chapter Notes - Chapter 9-10: Lone Pair, Chemical Polarity, Valence Electron
37 views5 pages
Document Summary
Chemical bonds are the forces that hold the atoms together in substances. A lewis electron-dot symbol consists of the symbol for the element surrounded by dots, one for each valence electron. An electron-configuration notation in which only the valence electrons of an atom of a particular element are shown, indicated by dots placed around the element"s symbol. Atomic symbols represent nuclei and inner-shell electrons. Dot-pairs or dashes between 2 atomic symbols represent electron pairs in covalent bonds. Dots adjacent to only one atomic symbol represent unshared electrons called. Lone pairs = they do not participate in bonding. Cations of most representative elements have no valence shell electrons shown in the lewis symbol. Lewis symbols of most monatomic anions show eight valence electrons. Ionic bonding results from electrostatic attraction between cations and anions. Formation if an ionic bond can be viewed as a transfer of electrons.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232