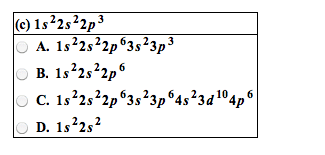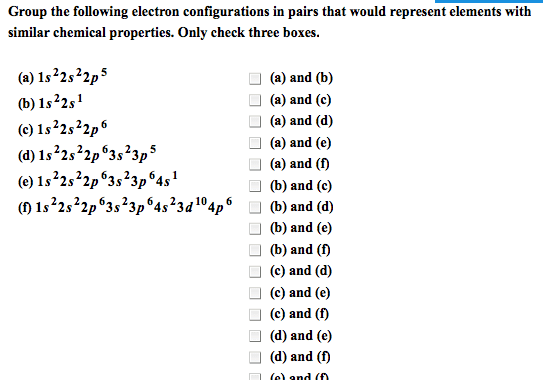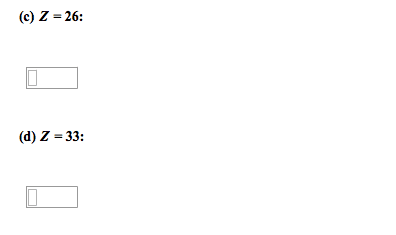CHEM 1220 Chapter Notes - Chapter 21: Ionic Radius, Lead, Electron Affinity
64 views4 pages
9 Apr 2015
School
Department
Course
Professor
Document Summary
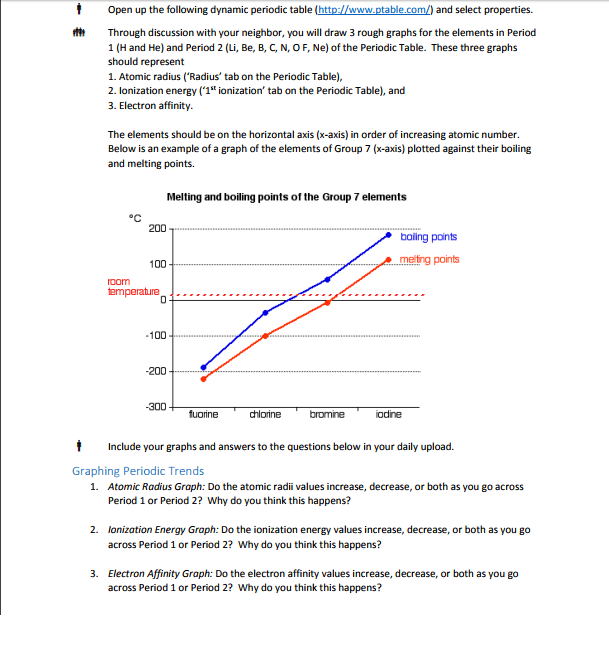
Chapter # 21: chemistry of main-group elements i: Periodic trends can be found in each group of elements in the periodic table. They have similar electronic configurations but not exactly the same chemical properties. Atomic properties that determine chemistry of element: (1) atomic radius (2) ionization energy (3) electron affinity (4) polarizability. Ionic radii p = (1. 60 x 10-19c)(z) (4/3) r3. The bond between metals and non-metals is ionic with a covalent character. Charge density (p) charge per unit volume in a cation. In a metallic bond, the higher the charge density of the cation, the greater its ability to distort the electron cloud around the anion. The greater the distortion of the electron cloud, the greater the covalent character of the bond. Alkali metals: z charge number r ionic radius relatively abundant on earth compounds were used since prehistoric times but elements were not isolated in pure form until about 200 years ago.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232